Voices of Biotech
Podcast: MilliporeSigma says education vital to creating unbreakable chain for sustainability
MilliporeSigma discusses the importance of people, education, and the benefits of embracing discomfort to bolster sustainability efforts.
July 1, 2010

Figure 1: ()
Selection of a CMO is critical to biologic drug development; and the primary selection criterion is experience. How do biotech companies measure experience? CMO experience can be measured in many ways — primary metrics include (1) total number of GMP processes developed by a CMO; (2) number of projects involving your strain; (3) number of projects involving your product type; (4) GMP production success rate; (5) total number of GMP lots produced; and (6) number of quality audits.”
Eurogentec’s biologics division is specialized in the manufacturing of biopharmaceuticals from microbial sources: bacteria, yeast, and biosafety level 2 (BSl-2) organisms. Eurogentec has manufactured according to GMP since 1994.
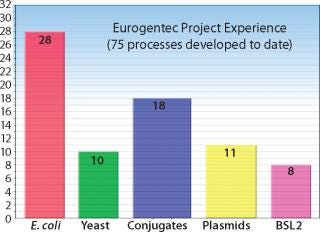
Figure 1: ()
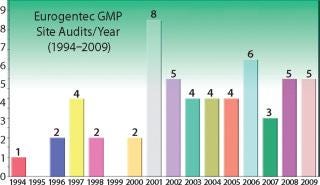
Figure 2: ()
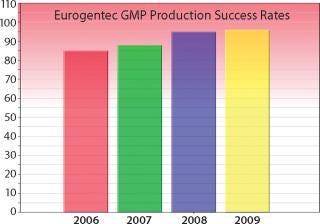
Figure 3: ()
Host Cell Experience: Microbial Experts
Eurogentec has expertise with E. coli, P. pastoris, H. polymorpha, S. cerevisiae, and numerous BSl-2 organisms.
Chemistry Experience: Conjugation Experts
Maleimide
Glutaraldehyde
Other chemistries available
Product Family Experience
Recombinant proteins
Plasmid DNA
Protein-PEG conjugations
Protein-protein conjugations
Protein-peptide conjugations
GMp Infrastructure
Multiproduct facility
Separate HVAC systems
GMP fermentation suites: 80 L, 150 L, 500 L fermenters
2 GMP purification suites
1 GMP sterile filtration suite
Process Development Strategies
FastTrack
OptiTrack
About the Author
Author Details
Dr. Pascal Bolon is biologics sales and marketing manager at Eurogentec SA, Rue du Bois Saint-Jean 5, 4102 Seraing, Belgium; +32 4- 366-6116; [email protected].
You May Also Like