Voices of Biotech
Podcast: MilliporeSigma says education vital to creating unbreakable chain for sustainability
MilliporeSigma discusses the importance of people, education, and the benefits of embracing discomfort to bolster sustainability efforts.

Single-use systems have become all the rage in the biotechnology industry, driven largely by the need to reduce cleaning requirements and associated time, validation, and cost factors associated with stainless vessels. A well-developed single-use system will ensure lower risk of contamination in manufacturing while eliminating the cleaning process and cleaning validation.
Providing Solutions
Advanced Scientifics provides specific solutions to process needs in fluid transfer and handling, offering flexibility in the design and componentry of individual systems.
Advanced Scientifics offers a wide assortment of transfer and filtration sets for most any application, providing sets that are used throughout the cell culture/fermentation process, from media or buffer transfer to seeding or harvest — to separation and purification — to final packaging. Advanced Scientifics offers more than 80 different tubing sizes and types, as well as more than 900 off-the-shelf connectors. As a leader in the design and manufacturing of single-use systems for the lifesciences and healthcare industries, Advanced Scientifics offers a multitude of solutions for flexible containers, as well as 3-D bags from 20 L and up, specific to customer specifications and tote dimensions.
Quality Process and Inspection
Advanced Scientifics is an ISO 13485:2003, FDA-registered manufacturer and uses a quality management system that is intertwined with its manufacturing system through the use of an enterprise resource planning (ERP) software. This high- quality ERP software improves operational efficiencies, customer satisfaction, and financial performance. From incoming raw material inspection through the entire manufacturing process to the final finished product, Advanced Scientifics’ ERP and quality management system is one of the industry’s fastest and strongest.
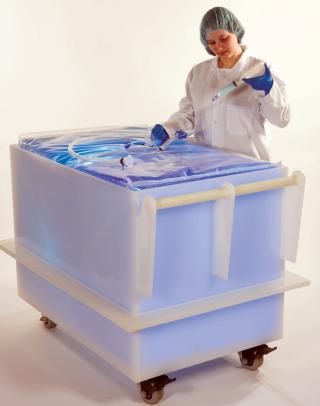

Specialized Product Delivery in 2–3 Weeks
After design, assembly, and packaging have been completed, Advanced Scientifics will further manage the sterilization of each single-use system. The company has validated this irradiation process to ensure that each product meets and exceeds the highest industry standards regarding sterility and validation, delivering a finished sterile product in two to three weeks.

About the Author
Author Details
Sheri Reber is marketing manager, 717-692-2104 x301, [email protected]; and Derek Masser is a lifesciences sales manager, 717-692-2104 x256, [email protected]; 163 Research Lane, Millersburg, PA 17061; www.advancedscientifics.com.
You May Also Like