Insider News
Insider Daily: BioProcess International Europe
MoreLab-grown fish offers a sustainable alternative to current seafood production methods but suffers many of the same manufacturing-based pain points as the biopharma industry, says cultivated fish firm BLUU.

Quality and compliance, customer service levels, financials, health and safety, and environmental sustainability are key to multi-modality manufacturing success, Novartis says.

With limited assets, startups must rely on CDMO partners. And working with the right ones is imperative even if it comes with a cost.
.jpg?width=700&auto=webp&quality=80&disable=upscale)
UCB says it built out its internal capabilities to create a process intensified AAV production process based on perfusion after finding limitations when working with external partners.
Latest eBooks
See AllSpecial Reports
In order to enhance access to life-saving medicines, the biopharmaceutical industry must prioritize more efficient therapeutic production. However, the environmental impact of manufacturing choices is facing increasing scrutiny. Sustainability opportunities within the biopharmaceutical sector encompass reducing the environmental footprint, addressing resource depletion, and effectively managing waste through repurposing and recycling. During a recent panel discussion, sustainability experts from Sartorius and their collaborators shared valuable insights on enhancing the sustainability of manufacturing processes. These insights encompass adopting single-use technologies, improving process efficiency, and fostering productive collaborations. This Special Report offers extracts from those enlightening discussions, along with relevant excerpts from Sartorius' white paper on sustainability in the biopharmaceutical industry.
Antibody–drug conjugates (ADCs) are complex therapeutics that offer a unique combination of selectivity and potency. Commercial demand for ADC therapeutics is projected to continue to increase while facing a very complex development, manufacturing, and supply chains challenges. Given the complexities of ADC development and manufacturing, there are clear advantages to partnering with a single contract development and manufacturing organization (CDMO) that offers a complete set of services. As ADCs continue to grab a larger share of clinical pipelines, it becomes increasingly essential to select a CDMO partner with the experience and expertise to accelerate timelines while minimizing risk. Fill out the form below to read this special report and learn more about ADC market, growth, challenges, and solutions to accelerate ADC pipelines to market.
Ask the Expert Webcasts
This webcast features: Heidi Jones, MBA, Market Development Manager, Chromatography Applications & Collaborations, Bio-Rad Laboratories.Endotoxins are common pyrogenic impurities in therapeutic protein preparations that make the next steps in the purification process very difficult. They are negatively charged complex aggregates of lipopolysaccharides released in large amounts from the outer membrane of gram-negative bacteria during mechanical cell lysis and death. Their presence in even minute concentrations in therapeutic protein preparations can cause septic shock, tissue injury, and even death.In research samples, endotoxins can hinder accurate data screening and immunological readouts. Endotoxins are extremely thermally stable and insensitive to pH changes, making their removal especially challenging. This webcast will present case studies using various resins that have been successfully used for endotoxin removal. Key Takeaways:
This webcast features: William H. Rushton, MS , Process Chromatography Scientist, Bio-Rad Laboratories. Recombinant adeno‐associated viruses (rAAV) are among the most promising vectors for long‐term gene transduction. These viruses have a high degree of safety, making them ideal for gene therapy applications. Significant progress has been made in improving rAAV vector production and purification. In this study, different chromatography workflow solutions were explored to purify rAAV8. Experimental conditions and data will be presented on using anion exchange (AEX) and mixed-mode chromatography to purify rAAV8. Key Takeaways: Thank you for your interest in this webcast. You may submit your request to view the webcast, and you will receive a confirmation email with log-in details.









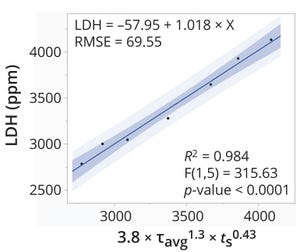

.png?width=300&auto=webp&quality=80&disable=upscale)