Voices of Biotech
Podcast: MilliporeSigma says education vital to creating unbreakable chain for sustainability
MilliporeSigma discusses the importance of people, education, and the benefits of embracing discomfort to bolster sustainability efforts.

Researchers have turned to Chinese hamster ovary (CHO)–based transient gene expression (TGE) as an alternative to CHO stable cell line production for early stage antibody development. Despite advances in transfection methods and culture optimization, most CHO-based TGE systems produce antibody titers (low mg/L) that are insufficient for full use in biotherapeutic development pipelines. MaxCyte electroporation is a universal, transient transfection platform with unmatched quality, flexibility, and scalability and the capacity to transfect up to 2 × 1011 cells in under 30 minutes. CHO transfection efficiencies and viabilities of >90% generate antibody titers over 400 mg/L within two weeks of transfection and can exceed 1 gram/L with optimization. MaxCyte electroporation can simultaneously create high-yield, stable CHO cell lines (>3 g/L productivity) in six to eight weeks using a simple selection protocol, thereby streamlining scale up to clinical-grade biomanufacturing.
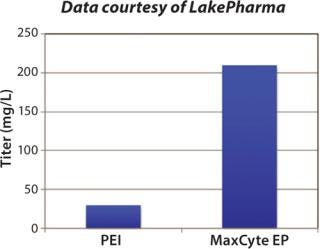
MaxCyte Electroporation Outperforms PEI
CHO transient transfection methods (including PEI and lipid-based reagents) generally provide antibody titers of 1–150mg/L with full optimization. Such methods can be limited in reproducibility, scalability, functional feasibility, and cost considerations. By contrast, MaxCyte electroporation is highly reproducible, simple to perform, and easily scaled without reoptimization. Direct comparison against PEI clearly shows the superior performance of MaxCyte electroporation. Secreted antibody titers were 7× higher using MaxCyte electroporation on Day 9 after CHO transfection (Figure 1).
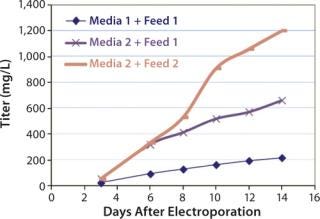
Antibody Titers >1 g/L with Media and Feed Optimization
Cell growth, nutrient depletion, and secreted protein titers are interrelated and significantly affected by media formulation and feeding strategies (Figure 2). Combining optimized media composition with a feed strategy in which nutrients are replenished more frequently generated antibody titers 3–6× that of unoptimized conditions. Antibody titers reached 1.2 g/L on Day 14 after electroporation, which translated into production of >3 grams of antibody from a single transfection. Even unoptimized culture conditions generated antibody titers >200 mg/L, demonstrating the high quality of MaxCyte transfection.
Conclusions
MaxCyte flow electroporation is a proven, high-efficiency means of CHO transient transfection for rapid production of gram-level quantities of antibodies, enabling its use in early phase candidate identification as well as later-stage studies requiring gram-level antibody quantities. Additionally, this single transfection technology can be harnessed to rapidly generate high-yield stable CHO cell lines, bridging the gap between early and late stage antibody development activities.
About the Author
Author Details
Krista Steger is a marketing consultant, Karen Donato is executive vice president of business development and marketing, Weili Wang is a scientist, and James Brady is director of technical applications at MaxCyte, Inc., 22 Firstfield Rd, Suite 110, Gaithersburg, MD 20878; 1-301-944-1700; [email protected]; www.maxcyte.com.
You May Also Like