Content Spotlight
Podcast: MilliporeSigma says education vital to creating unbreakable chain for sustainability
MilliporeSigma discusses the importance of people, education, and the benefits of embracing discomfort to bolster sustainability efforts.
Astellas will codevelop immunostimulatory antibody-drug conjugates (iADCs) for three biological targets through a partnership with Sutro Biopharma.
Japanese biopharma firm Astellas will pay Californian biotech Sutro $90 million but could pay a further $422.5 million in development, regulatory and commercial milestones for each of the three undisclosed oncology candidates.
While antibody-drug conjugates (ADCs) have established themselves on the commercial front, with 12 approved in the US, Sutro says its engineered iADCs are a potential groundbreaking modality in the treatment of cancers for which no broadly effective therapy is currently available.
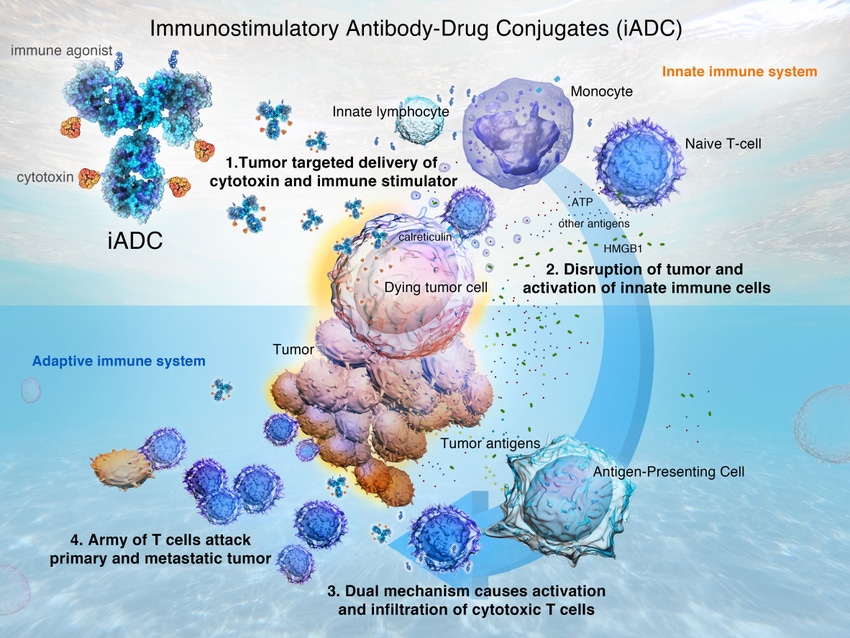
Method of action for iADCs
“Similar to ADCs, iADCs combine an antibody with a cytotoxic small molecule compound to induce immunogenic cell death. But iADCs, which represent a next-generation anti-cancer modality, have an added component, which is the immune modulating small molecule contained on a single antibody,” Trevor Hallam, Sutro’s CSO told BioProcess Insider.
The modality works by inducing immunogenic cell death in conjunction with immune modulation and are designed to both kill the tumor locally and prime a systemic anti-tumor immune response, he continued.
“The dual mechanism of action is expected to boost anti-cancer action and has the potential to turn cold tumors hot and bring new drug therapies to patients who do not respond to existing therapies.”
Under terms of the arrangement, Sutro will engage in research and preclinical studies to identify candidate compounds and Astellas will pursue clinical development using its global R&D and commercialization capabilities in the area of antibodies and the small molecular components.
“We’re very excited about this partnership and working together as Astellas is a leading biopharmaceutical company – particularly within the oncology space – that has built strong global R&D and commercialization capabilities in antibodies and small molecular compounds, which we believe will be instrumental to the long-term success of these novel drugs,” said Hallam.
You May Also Like