Content Spotlight
Podcast: MilliporeSigma says education vital to creating unbreakable chain for sustainability
MilliporeSigma discusses the importance of people, education, and the benefits of embracing discomfort to bolster sustainability efforts.
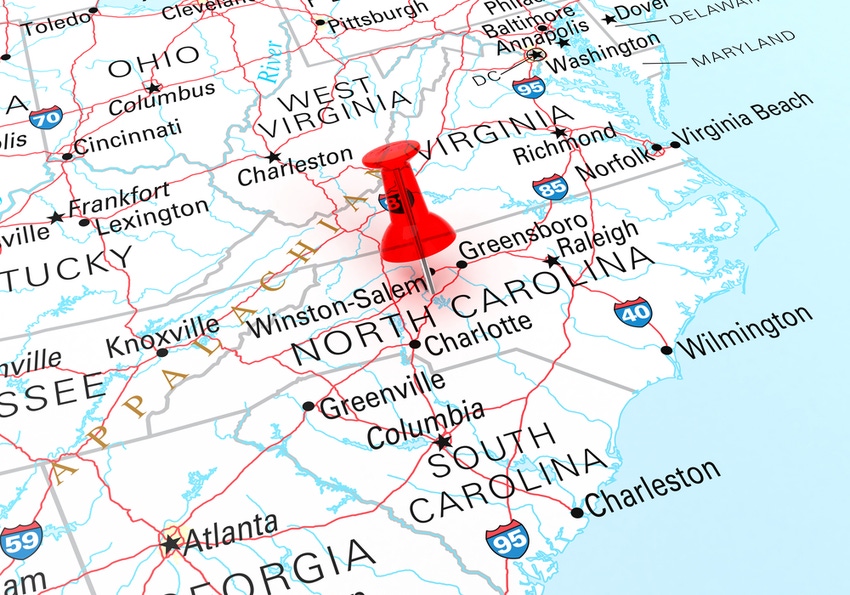
The Research Triangle Park facility will significantly expand Fujifilm Irvine Scientific’s cell culture media capacity to feed the projected double-digit growth in demand from industry.
The planned 250,000 square-foot facility – construction of which is scheduled to start in spring 2023– will produce animal component-free, dry powder, and liquid media for biomanufacturing customers.
The site in Research Triangle Park (RTP), North Carolina will be the latest in Fujifilm Irvine Scientific’s cell culture media network, joining production sites in Santa Ana (California), Tokyo (Japan), and Tilburg (The Netherlands).
Image: DepositPhotos/
klenger
When operational in 2025, the site will significantly triple current capacity for liquid cell culture media (through an additional 3,300,000 L/year), while also adding 800,000 Kg/year for dry powder, and 40,000 L/day of Water for Injection (WFI).
A spokesperson told us the decision to invest $188 million in another plant comes from the forecasted demand for cell culture media, which according to a 2020 BioPlan Associates report, is projected to grow at a CAGR of 10-12% through 2031.
“This is due to increasing demand for bioproduction services and advanced therapies such as cell and gene therapies,” we were told. “In addition, Fujifilm Irvine Scientific is increasing its market share. The Company projects it will need the additional capacity provided by the RTP facility by 2027.”
Fears of a slowdown post-pandemic were also abated, with the spokesperson telling BioProcess Insider: “Demand was calculated with the drop off in COVID related orders factored in. The Company is confident the demand is there based on forecasts of approved therapies and new therapies in early phases of development.”
Approximately 100 positions will be created through the investment.
Parent company Fujifilm Corporation is also behind another major capex project in North Carolina; the ¥200 billion ($1.4 billion) biomanufacturing site in Holly Springs, located about 10 miles southwest of Raleigh, being constructed by contract development and manufacturing organization (CDMO) Fujifilm Diosynth Biotechnologies (FDB).
Both subsidiaries are independent, and the latest investment was not designed to support the facility under construction by FDB. However, “it will ensure a secure supply chain, and rapid, reliable delivery of products through the development of extensive regional supply networks, critical to supporting cell and gene therapies,” we were told.
“RTP in North Carolina is an ideal second location in the United States. Regional partners such as the Research Triangle Foundation, Wake County Economic Development, Economic Development Partnership of NC, and the NC Department of Commerce have built a community for life science companies that is rich in skilled talent, and resources to facilitate business.
“The region is popular with life science companies including many current and potential customers of Fujifilm Irvine Scientific. The proximity of the new facility to customers on the east coast will also provide them with efficient and faster shipping.”
You May Also Like