Content Spotlight
Podcast: MilliporeSigma says education vital to creating unbreakable chain for sustainability
MilliporeSigma discusses the importance of people, education, and the benefits of embracing discomfort to bolster sustainability efforts.
March 4, 2020
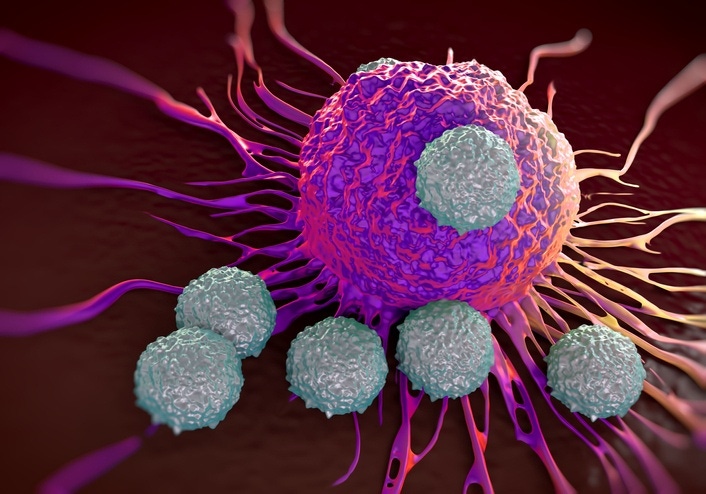
The acquisition of monoclonal antibody developer Forty Seven complements Kite’s CAR-T therapy franchise in targeting cancers, says Gilead.
Under deal terms announced Monday, Gilead will pay $95.50 (€85.50) for each share of Menlo Park, California-based Forty Seven, totaling $4.9 billion.
Forty Seven’s lead CD47-blocking drug, an antibody called magrolimab, is being tested in combination with chemotherapy in separate studies evaluating it as a treatment for two types of blood cancer: a group of disorders called myelodysplastic syndromes (MDS) and acute myeloid leukemia (AML).
Image: iStock/royaltystockphoto
The Forty Seven drug is being developed for additional cancers including non-Hodgkin lymphoma and diffuse B-cell lymphoma.
Speaking on a conference call Monday, Gilead CEO Daniel O’Day said that both AML and MDS have a high unmet need. As a cancer treatment that is not a cell therapy, Forty Seven’s assets complement the cell therapy franchise that Gilead added with its 2017 acquisition of Kite Pharma, he added.
“This is in the sweet spot, I think, of what you need to do to be successful in oncology,” O’Day said. “We will not be afraid to invest as we see data and as we see signals in the data to move fast and be first with combinations with this molecule.”
Gilead reported that it ended 2019 with $25.8 billion in cash and marketable debt securities. When the company released its 2019 fourth quarter and full year financial results last month, O’Day announced a goal to introduce 10 “transformative medicines” in the next 10 years. Reaching that goal would require partnerships and small-to-medium-sized acquisitions.
Gilead characterizes the Forty Seven acquisition as a medium-sized deal. O’Day said Gilead is looking to acquire other companies that have clinical-stage assets supported by strong science.
Gilead expects to complete the deal in the second quarter.
You May Also Like