Voices of Biotech
Podcast: MilliporeSigma says education vital to creating unbreakable chain for sustainability
MilliporeSigma discusses the importance of people, education, and the benefits of embracing discomfort to bolster sustainability efforts.
 As advanced therapies, including regenerative medicines, progress toward commercialization and market approval, early warnings from key opinion leaders (1, 2) regarding the importance of better understanding quality target product profiles (QTPPs) and critical quality attributes (CQAs) of such products have resounded ever louder (see the “Terminology” box for definitions). Costly late-stage delays, redirections, and even abandonment of clinical programs can be linked to quality issues associated with inadequate understanding of process and product. Therefore, a review of the benefits of a quality by design (QbD) approach to optimizing and informing the production processes is needed to reinforce the necessity of building quality into a process and to provide a framework in which early indications of quality issues can be ascertained (3).
As advanced therapies, including regenerative medicines, progress toward commercialization and market approval, early warnings from key opinion leaders (1, 2) regarding the importance of better understanding quality target product profiles (QTPPs) and critical quality attributes (CQAs) of such products have resounded ever louder (see the “Terminology” box for definitions). Costly late-stage delays, redirections, and even abandonment of clinical programs can be linked to quality issues associated with inadequate understanding of process and product. Therefore, a review of the benefits of a quality by design (QbD) approach to optimizing and informing the production processes is needed to reinforce the necessity of building quality into a process and to provide a framework in which early indications of quality issues can be ascertained (3).
The cell and gene therapy industry has experienced incredible growth and achieved significant milestones in recent years, including the rapid progress from early clinical success to market approval of chimeric antigen receptor (CAR) T-cell therapies for hematologic cancers (4, 5). These advances have been catalysts for spectacular capital investments from venture-capital firms and pharmaceutical companies alike, including Gilead’s US$11.9 billion acquisition of Kite Pharma (6) and Celgene’s $9 billion bid for Juno Therapeutics (7), along with other substantial transactions (8).
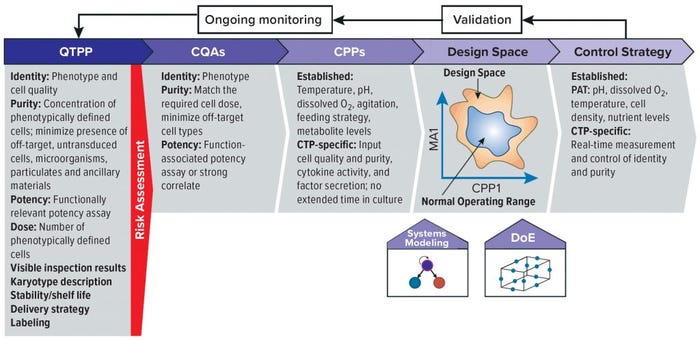
Figure 1: The first step in applying quality by design (QbD) is to define a quality target product profile (QTPP) — the characteristics that assure quality, safety, and efficacy. Second, quality attributes that are critical for meeting the QTPP are determined through risk assessment. Third, the critical process parameters (CPPs) and materials attributes (MAs) that affect critical quality attributes (CQAs) are identified, and their effects on those attributes are quantified in a design space. Fourth, a control strategy is developed to ensure that CPPs remain within the “normal operating range” that ensures manufacture of high-quality product. Fifth, the process is validated in a manufacturing facility at scale, and it is monitored continually during manufacturing runs and improved as knowledge about the process increases. This figure includes both established analyses common to QbD processes for all therapeutic agents and those specific to cell therapy processes, but some elements could have parameters that are not represented here. —adapted from (8)
Concurrently, some thought leaders have made efforts to share lessons learned from innovations in biopharmaceutical processing and control to expedite the path of these unique therapeutics for addressing unmet medical needs (9), particularly through the iterative process of QbD as shown in Figure 1 (10). The QbD process was founded in guidance documents released in 2006 for addressing pharmaceutical development (11), quality risk management (12) and quality systems (13) in response to increasing complexity of biologic products. The value of this approach is recognized by regulators worldwide. Three years ago the European Medicines Agency (EMA) and the US Food and Drug Administration (FDA) reported on a five-year joint pilot program to assess marketing-authorization applications for products developed using QbD processes (14). The market benefits of a harmonized approach have been evident in both product knowledge and expedited approvals (15, 16).
Given the risks of failure in later stages of clinical development, a rational and pragmatic approach to defining product specifications — driven by quality but also flexible enough to accommodate broad measurement ranges and expanding efficacy data — is particularly prudent for personalized medicines. This prompts two questions: What challenges have kept such an approach from being leveraged for cell and gene therapies? Can purposeful refinement of QbD tenets offer the same advantages to this early stage industry that it has to other biological products?
Adoption Challenges and Potential Adjustments
Fundamental issues that must be addressed in adopting a QbD framework for advanced therapies include
the personalized nature of individual therapies, for which variability of cellular materials across patient populations and final products can be immense, even for controlled processes
a limited understanding of the mechanisms of action (MoAs) and their links to measurable product attributes — e.g., presence of cytokines and/or cell markers
the complexity of manufacturing processes for which comprehensive understanding of platforms and the potential interactions between numerous variables are not well understood
a lack of robust process analytical technologies (PATs) for monitoring critical process parameters (CPPs) that are in-line or miniaturized for cell-expansion and other associated processes
the diversity of product types and product-specific questions that prohibits application of technology and know-how directly from one manufacturing process or product to another.
Many cell/gene therapies are personalized by nature, so a systematic, data-driven approach of connecting CPPs to CQAs can be hindered by donor variability (17), sample-volume availability, and sample quantities (18). In addition direct correlation between CPPs and patient outcome has proven problematic (19), suggesting that the determination of relevant CPPS and CQAs will be more complicated. Although the conventional biopharmaceutical practice of overlaying replicate runs in an effort to establish an aspirational “golden batch” standard might not apply here, aggregating parameters and outcomes from multiple patients could offer better understanding of a process design space within which products can be considered to be safe and efficacious (Figure 1) and honed through life-cycle management. Such a broadened interpretation that includes evolving input to release criteria could allow for unavoidable attributes of a “living” product (e.g., differences in culture processes or timelines caused by growth dynamics or other aspects) and be tightened continually as patient numbers increase over time.
Quality By Design Terminology |
Quality by design: a scientific, risk-based framework for process design based on relating product and process attributes to product quality Quality target product profile: the properties of the end product desired for clinical use Critical process parameter: variable elements of the production process that influence critical quality attributes Material attribute: variable elements of the input materials that influence critical quality attributes Design of experiments: control of experiments to identify system response to inputs Design space: range of variability in critical process parameters and material attributes to achieve critical quality attributes Control strategy: method of maintaining critical process parameters and material attributes within a design space Process analytical technology: a system to design, measure, monitor, and control critical process parameters |
Many cell/gene therapies target complex pathways and biological systems such as immunity, in which active discovery remains a regular occurrence. So it is understandable that a definitive MoA might not always be apparent, potentially making its conjunction with QTPPs, CQAs, and CPPs of such products challenging to achieve. That lack of MoA clarity should neither confine the development process nor restrict clinical progress of effective therapies. Historical experience has proven that model organisms do not always indicate direct human clinical success or failure (20), so delays for further research cannot be relied on to yield fruitful evidence. Instead, the results should offer a well-considered opportunity to build on original clinical hypotheses and design of experiments (DoE) with additional data points that could elucidate potential markers or parameters and provide practical insight into those that may be rejected as uninformative (and thus unnecessary for future lot testing).
PAT devices for monitoring pH, dissolved oxygen (DO), glucose concentration, cell density, and other biopharmaceutical CPPs have made great strides in miniaturization and integration. However, cell and gene therapies regularly require higher-level or destructive detection devices, analytics, and separation technologies such as flow cytometry, metabolomics, selection, and similar techniques — often with reduced intra- and interassay reproducibility (21). Access and implementation costs for such platforms, lengthy analysis times, and complexities in data interpretation make the methods difficult to use in controlled loops or effective management strategies.
Note, too, that every sampling event that is not a closed, in-line, or on-line interaction puts precious autologous products at risk of contamination. The integral risk assessments of a QbD approach should identify at- or off-line measurements that would be valuable without being detrimental, and the resulting data again could heighten an otherwise limited understanding of the process dynamics and differences. Another option, when statistically driven DoE is not possible because patient samples are unavailable or the number of potential variables is onerous, would be to develop more predictive mathematical modeling of these biological systems that can offer relevant readouts as supporting data (22).
Benefits of Implementation
If broader QbD understandings accounting for the complexity and limitations of cell and gene therapies could become the norm, the potential benefits to product knowledge and innovation could far outweigh the risks involved in implementation. Two fundamental results would be
improved depth and development of augmented chemistry, manufacturing, and controls (CMC) filing packages
accessibility and feasibility of late-stage introductions of technology innovations.
Late-stage and even market-authorized products such as Novartis’s Kymriah (tisagenlecleucel) and bluebird bio’s Zynteglo (autologous CD34+ cells encoding βA-T87Q-globin gene) have experienced production challenges that led to launch delays (23) and reduced earnings caused by out-of-specification (OoS) products (24). This is not to say that QbD would resolve all such complicated, convoluted, and multifactorial problems. But enhanced understanding of the products and their manufacturing processes could offer a route to proactive improvements and informed decision-making when OoS events do occur. Increased control and product knowledge have been foundational for regulatory advancements in protecting patient safety for many biopharmaceuticals, and advanced therapy products should be no different.
Global Advanced-Therapy |
Corporate Partnerships/Collaborations
Follow-on Financing
Venture Financing
Mergers and Acquisitions
|
The largest potential impact could be to enable introduction and experimentation on comparable, improved materials and novel processing devices and innovations for late-stage and approved processes with limited regulatory oversight, documentation, and review times — as came with the advent of the “well-characterized biologicals” paradigm. If a well-characterized QTPP is based on efficacy outcomes, with precise CQAs and historically documented CPP control over product and process design spaces, then changes to the ancillary materials, equipment, and technologies used in manufacturing should be acceptable and even advisable to maintain a “best-in-class” product. Expanded process flexibility could empower CoG-saving measures and facilitate timely quality upgrades for raw materials. It also would enable equipment innovations — and more important, burgeoning fit-for-purpose technologies answering the industry’s call to address imperative needs related to small sample sizes, PAT affordability, sterility, and connectivity — that could offer stepwise benefits immediately rather than decades into the future.
The Path Forward
“Living” therapies require “living” strategies of development, commercialization, and regulation that can change and develop as new information arrives and as novel technologies are discovered. Putting creative thinking into the application of QbD principles to cell and gene therapy products is by far the best alternative to ignoring their value or missing out on their potential benefits. With efforts made toward expedient yet safe therapeutic offerings for unmet clinical needs, thorough risk assessments and visionary problem solving will be critical to success of the advanced-therapeutics industry. Building on the pharmaceutical industry’s motto of “fail fast,” this market must become thoughtfully refined and embody the practical amendment: “Correct faster.”
References
1 Preti B, et al. Mapping Success for Commercial Cell Therapy Manufacturing. BioProcess Int. 13(9) 2015: S33–S39.
2 Joydeep B, Ludlow JW. Cell-Based Therapeutic Products: Potency Assay Development and Application. Regen. Med. 9(4) 2014: 497–512; https://doi.org/10.2217/rme.14.25.
3 Rathore AS, Winkle H. Quality By Design for Biopharmaceuticals. Nat. Biotechnol. 27(1) 2009: 26–34; https://doi.org/ 10.1038/nbt0109-26.
4 Fesnak AD, June CH, Levine BL. Engineered T Cells: The Promise and Challenges of Cancer Immunotherapy. Nat. Rev. Cancer 16(9) 2016: 566–581; https://doi.org/10.1038/nrc.2016.97.
5 FDA Approves Axicabtagene Ciloleucel for Large B-Cell Lymphoma. US Food and Drug Administration: Rockville, MD, 25 October 2017; https://www.fda.gov/drugs/resources-information-approved-drugs/fda-approves-axicabtagene-ciloleucel-large-b-cell-lymphoma.
6 Gilead Sciences to Acquire Kite Pharma for $11.9 Billion. Gilead Sciences, Inc.: Foster City, CA, 28 August 2017; https://www.gilead.com/news-and-press/press-room/press-releases/2017/8/gilead-sciences-to-acquire-kite-pharma-for-119-billion.
7 Celgene Corporation to Acquire Juno Therapeutics, Inc. Celgene Corporation: Summit, NJ, 22 January 2018; https://www.celgene.com/newsroom/cellular-immunotherapies/celgene-corporation-to-acquire-juno-therapeutics-inc.
8 2018 Annual Report. Alliance for Regenerative Medicine: Washington, DC, 2018; https://alliancerm.org/publication/2018-annual-report.
9 Campbell A, et al. Concise Review: Process Development Considerations for Cell Therapy. Stem Cells Transl. Med. 4(10) 2015: 1155–1163; https://doi.org/10.5966/sctm.2014-0294.
10 Lipsitz YY1, Timmins NE2, Zandstra PW. Quality Cell Therapy Manufacturing By Design. Nat. Biotechnol. 34(4) 2016: 393-400; https://doi.org/10.1038/nbt.3525.
11 CBER/CDER. Guidance for Industry: Q8 Pharmaceutical Development. US Food and Drug Administration: Rockville, MD, May 2006; https://www.fda.gov/media/71524/download.
12 CBER/CDER. Guidance for Industry: Q9 Quality Risk Management. US Food and Drug Administration: Rockville, MD, June 2006; https://www.fda.gov/media/71543/download.
13 CBER/CDER. Guidance for Industry: Q10 Pharmaceutical Quality System. US Food and Drug Administration: Rockville, MD, September 2006; https://www.fda.gov/media/71553/download.
14 EMA 213746/2017. EMA-FDA Pilot Program for Parallel Assessment of Quality By Design Applications. European Medicines Agency: London, UK, March 2011; www.ema.europa.eu/docs/en_GB/document_library/Other/2017/04/WC500225533.pdf.
15 Zurdo J, et al. Early Implementation of QbD in Biopharmaceutical Development: A Practical Example. BioMed Res. Int. 17 May 2015; https://doi.org/10.1155/2015/605427.
16 Ohage E, et al. QbD Implementation and Post Approval Lifecycle Management (PALM). Biologicals 44(5) 2016: https://doi.org/10.1016/j.biologicals.2016.06.007.
17 Silverman LI, et al. Identifying and Managing Sources of Variability in Cell Therapy Manufacturing and Clinical Trials. Regen. Eng. Transl. Med. 5, 2019: 354–361; https://doi.org/10.1007/s40883-019-00129-y.
18 Banerjee MN, Bolli R, Hare JM. Clinical Studies of Cell Therapy in Cardiovascular Medicine: Recent Developments and Future Directions. Circ. Res. 123, 2018: 266–287; https://doi.org/10.1161/CIRCRESAHA.118.311217.
19 Tyagarajan S, Spencer T, Smith J. Optimizing CAR-T Cell Manufacturing Processes During Pivotal Clinical Trials. Mol. Ther. Meth. Clin. Devel. 16, 2020: 136–144; https://doi.org/10.1016/j.omtm.2019.11.018.
20 Leenaars CHC, et al. Animal to Human Translation: A Systematic Scoping Review of Reported Concordance Rates. J. Transl. Med. 17, 2019: 223; https://doi.org/10.1186/s12967-019-1976-2.
21 Masri F, Cheeseman E, Ansorge S. Viral Vector Manufacturing: How to Address Current and Future Demands? Cell Gene Ther. Ins. 5(5) 2019: S949–S970; https://doi.org/10.18609/cgti.2019.104.
22 Kirouac DC, Zandstra PW. The Systematic Production of Cells for Cell Therapies. Cell Stem Cell 3(4) 2008: 369–381; https://doi.org/10.1016/j.stem.2008.09.001.
23 Palmer E. Bluebird Bio Delays Zynteglo Launch As Manufacturing Trips Up Another Gene Therapy. Fierce Pharma 19 June 2019; https://www.fiercepharma.com/manufacturing/bluebird-bio-s-delays-zynteglo-launch-as-manufacturing-trips-up-another-gene-therapy.
24 Sagonowsky E. Novartis Building Out Cell and Gene Therapy Platform That’ll Lead the Pack, CEO Promises. Fierce Pharma 7 January 2019; https://www.fiercepharma.com/pharma/novartis-cell-and-gene-therapy-work-about-expanding-game-board-treatments-ceo.
Corresponding author Kim Bure is chief commercial officer of AveriCELL in Northborough, MA; [email protected]. Yonatan Lipsitz is team lead in cell therapy process development at Sana Biotechnology, Inc. Mark Lowdell is chief scientific officer of INmuneBio and a clinical scientist in the Institute of Immunity and Transplantation at University College London. Ivan Wall is a professor in regenerative medicine (cell and gene therapy bioprocessing) at Aston University in Birmingham, UK. And Peter Zandstra is director of Michael Smith Laboratories; professor at and director of the School of Biomedical Engineering at the University of British Columbia in Vancouver, BC, Canada; cofounder and chief scientific officer of the Center for Commercialization of Regenerative Medicine (CCRM); cofounder and chief technology officer of Excellthera; and cofounder of Notch Therapeutics.
You May Also Like