Voices of Biotech
Podcast: MilliporeSigma says education vital to creating unbreakable chain for sustainability
MilliporeSigma discusses the importance of people, education, and the benefits of embracing discomfort to bolster sustainability efforts.
October 14, 2020
Date: Oct 14, 2020
Duration: 20 Min
Sponsored by Cygnus Technologies
This webcast features: David Cetlin, Senior Director, R&D, Cygnus Technologies
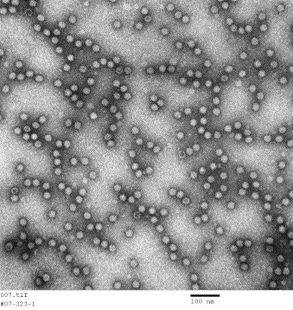
As viruses can arise during the manufacture of biopharmaceuticals, regulatory agencies require viral clearance validation studies for each biopharmaceutical prior to approval. These studies are typically conducted in biosafety level (BSL)-2 facilities and require large capital and human resources. Due to these hurdles, process knowledge pertaining to viral clearance is limited during development and characterization.
The use of an accurate, economical, and quantifiable non-infectious viral surrogate would enable downstream purification scientists to study viral clearance throughout process development.
This webinar will explore the use of a BSL-1 compatible, non-infectious minute virus of mice (MVM) particles to predict viral clearance results over a wide variety of process development applications such as anion-exchange (AEX) design of experiments (DoE) study, ion-exchange (IEX) high-throughput screening, and downstream purification of an adenoassociated virus (AAV)-based viral vector.
Takeaways:
Viral clearance prediction is possible through the use of non-infectious surrogates such as MVM mock virus particles (MVM-MVPs) that mimic the physiochemical properties of live mammalian viruses.
MVM-MVPs can be applied to downstream efforts such as DoE and high-throughput screening and help facilitate process development/optimization.
AAVX resin (Thermo Fisher Scientific) was shown to be an effective viral clearance step within an AAV downstream purification process, and comparative MVM versus MVM-MVP data showed favorable prediction through the use of the non-infectious surrogate.
Watch the recorded webcast now.
You May Also Like