Sponsored by Nuvonis
Newcastle disease is an extremely infectious condition among domesticated poultry and other avian species. Its high morbidity and mortality rates among infected birds give the disease significant economic importance. Thus, many commercially available vaccines based on live or inactivated virions are used globally to protect against Newcastle disease infection.
The causative agent is Newcastle disease virus (NDV), which belongs to the Paramyxoviridae family. NDV is a single-stranded, negative-sense, enveloped RNA virus of avian origin that is highly attenuated in humans and other primates because of strong host-range restriction (1). Attenuated NDV has been evaluated as a vector for vaccines against SARS-CoV-2 (2–4), Ebola (5), H5N1 influenza (6), West Nile (7), and simian immunodeficiency viruses (8). Oncolytic NDV vectors also hold much promise for immunotherapies against various cancers (9, 10).
Whether for vaccines or viral-vector therapies, NDV particles must meet certain criteria for yield, purity, and concentration. Previously, we have described a highly effective process for purification of influenza virus produced in Vero cells (11). Based on CIMmultus SO3 (sulfonate) monolith columns, the process yielded excellent recovery and impurity removal and enabled good manufacturing practice (GMP) scale-up to produce clinical-grade material. Here, we describe our adaptation of the process to NDV purification and thus demonstrate the broad applicability of SO3 monolith technology.
Avian Cell Lines and Monolith Chromatography for Vaccine Manufacture
Many NDV-based vaccines and viral vectors are still produced in traditional substrates such as embryonated chicken eggs or primary chicken embryo fibroblasts (CEFs). However, CEFs are not considered to be a robust manufacturing system, and they require large numbers of specific-pathogen–free (SPF) embryonated eggs to generate primary cell cultures (12). CEFs are susceptible to microbial contamination, and heterogeneity in their starting material leads to variability in NDV yield and quality. Avian cell lines have become popular alternatives to egg and primary-fibroblast technologies for human and veterinary vaccine manufacturing. Examples include DF-1 (chicken), AGE1 (duck), QOR (quail), and EB66 (duck) cell lines (13).
A Novel Avian Suspension-Cell Line: Nuvonis recently introduced the CCX.E10 quail-derived cell line (14). It offers several advantages over other current avian cell lines. Because it was developed without the use of foreign genes and viral sequences, it is not a genetically modified organism (GMO) cell substrate. The cell line grows in suspension culture, but an anchorage-dependent version is available for companies that want to use existing production infrastructure (e.g., roller bottles, stacked-tray systems, and microcarriers). The CCX.E10 line also grows in commercially available serum-free and animal-component–free media.
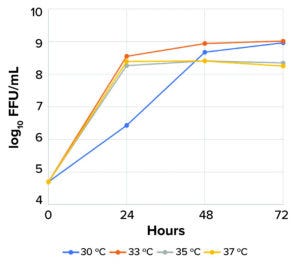
Figure 1: Upstream optimization of Newcastle disease virus (NDV) LaSota strain infectious titer, based on time after infection and temperature of incubation (FFU = focus-forming units).
When infected with NDV, CCX.E10 cells produce high viral titers. Nuvonis has demonstrated that the LaSota strain of NDV can be grown to 9 log10 focus-forming units (FFU)/mL at low multiplicity of infection (MoI) within 24–48 hours (Figure 1). This result exemplifies the cell line’s potential as a platform technology for a broad range of NDV products in human and veterinary medicine. Both GMP master and working cell banks (MCBs, WCBs) of the CCX.E10 cell line are fully characterized according to guidelines from the International Council for Harmonisation of Technical Requirements for Pharmaceuticals for Human Use (ICH) and can be used immediately for GMP-grade production.
Below, we describe our optimization of NDV LaSota production using CCX.E10 suspension cells. After culture, we collected high-titer harvests and purified them using CIMmultus SO3 monoliths. Our data confirm the applicability of monolith technology for efficient manufacturing of highly pure NDV preparations.
Materials and Methods
Production: CCX.E10 suspension cells were cultivated at 37 °C in a modified FreeStyle 293 expression medium (Thermo Fisher Scientific) in single-use spinner flasks ranging from 500 mL to 3 L (Corning). Cell populations doubled about once per day depending on the cell density. To evaluate NDV growth, CCX.E10 cells were infected at a density of ~106 cells/mL (MoI 0.05) and cultivated for three days at 30–37 °C. Infectious titers were analyzed using a focus-forming assay (FFA) in Vero cells, essentially as described (15). Harvests were clarified by centrifugation for 15 minutes at 4,400g and subsequently treated for two hours at room temperature with Benzonase endonuclease
(20 U/mL, MilliporeSigma) and 2 mM MgCl2.
Purification: We washed 1-mL CIMmultus SO3 columns and the chromatographic system with water and sanitized them using 1 M NaOH for at least an hour. After a wash with 10 column volumes (CV) of water, column pH was equilibrated using 10 CV of 1 M ammonium acetate, and another water wash (10 CV) was done. Conditioning was performed with 50 mM HEPES buffer and 200 mM saccharose (pH 7.0, 10 CV); 50 mM HEPES, 200 mM saccharose, and 2 M NaCl
(pH 7.0, 10 CV); and finally, 50 mM HEPES and
200 mM saccharose (pH 7.0, 20 CV).
As with influenza A and B purification (11), only dilution of Benzonase-treated clarified harvest (HCB) material is required before loading NDV onto an SO3 column. We diluted 100 mL of NDV harvest in a 1:1 ratio with equilibration buffer (50 mM HEPES and
200 mM saccharose, pH 7.0), and the SO3 column was loaded at 10 CV/min. We washed the monolith with 50 mM HEPES, 200 mM saccharose, and 75 mM NaCl (pH 7.5) at the same flow rate for 100 CV. Then, we eluted the material at a linear gradient over 50 CV from 0% to 100% of 50 mM HEPES, 200 mM saccharose, and 2 M NaCl (pH 7.5), with a hold step at 100% for 30 CV at a flow rate of 5 CV/min. After elution, cleaning-in-place (CIP) followed, using 1 M NaOH and 2 M NaCl.
Fractions were analyzed for several parameters. In addition to measuring ultraviolet (UV) absorbance at 260 nm and 280 nm, we confirmed the presence of large particles in the eluate using multiangle light scattering (MALS). Infectious titer was determined using an FFA (15). Total protein and DNA concentrations were assessed with PicoGreen and Bradford assay kits from Thermo Fisher Scientific and Bio-Rad Laboratories, respectively. To assess Benzonase concentrations, an enzyme-linked immunosorbent assay (ELISA) from MilliporeSigma was used, and nanoparticle-tracking analysis (NTA, NS300 instrument, Malvern Panalytical) was performed to determine particle-size distribution.
High Recovery and Purity
Replication of NDV LaSota in CCX.E10 cells at different temperatures showed that virus kinetics and final titer strongly depend on temperature. The highest infectious titers (9 log10 FFU/mL) were obtained at 30 °C and 33 °C after 48–72 hours of infection (Figure 1).
Virus purification starts with a 1:1 dilution of HCB to lower loading-material conductivity, which enables binding of NDV virions to the CIMmultus SO3 column. The downstream process from sample preparation through purification lasts less than an hour.
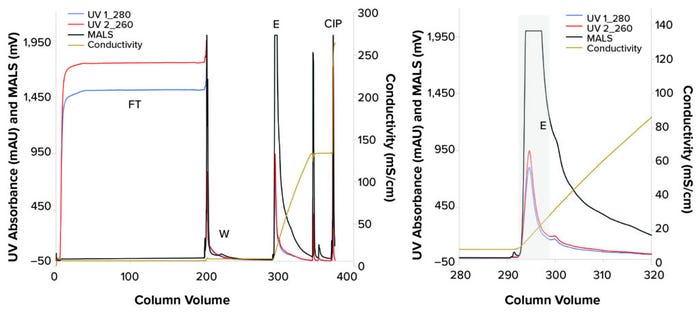
Figure 2: Purification of Newcastle disease virus (NDV) using a 1-mL CIMmultus SO3 monolith; the left panel provides the complete chromatogram, and the right panel focuses on the elution step. (CIP = cleaning in place, E = elution fraction, FT = flowthrough, MALS = multiangle light scattering, UV = ultraviolet, W = wash).
During flowthrough (FT), high UV absorbance signals indicated strong protein and DNA impurity removal, and a low MALS signal suggested the absence of large particles in the FT fraction (Figure 2). We verified that result with an FFA, which showed 0.1% of infectious NDV in the FT fraction.
After column washing, all three signals detected components eluting from the column, with MALS indicating a high concentration of large particles in the elution fraction (Figure 2). The main elution fraction was collected for 6.3 CV, resulting in 75% recovery of infectious particles and 99.0% depletion of proteins and 99.9% removal of DNA impurities (Figure 3a). Benzonase concentration in the eluate was below the instrument limit of quantitation (LoQ, 0.2 ng/mL). Thus, >99.8% of Benzonase reagent was depleted. Size distribution of eluted viral particles was more uniform compared with that of the load material, with mode particle sizes of 188.5 ± 1.9 nm and 168.9 ± 8.6 nm, respectively (Figure 3b). No aggregates were observed. 260 nm and 280 nm, we confirmed the presence of large particles in the eluate using multiangle light scattering (MALS). Infectious titer was determined using an FFA (15). Total protein and DNA concentrations were assessed with PicoGreen and Bradford assay kits from Thermo Fisher Scientific and Bio-Rad Laboratories, respectively. To assess Benzonase concentrations, an enzyme-linked immunosorbent assay (ELISA) from MilliporeSigma was used, and nanoparticle-tracking analysis (NTA, NS300 instrument, Malvern Panalytical) was performed to determine particle-size distribution.
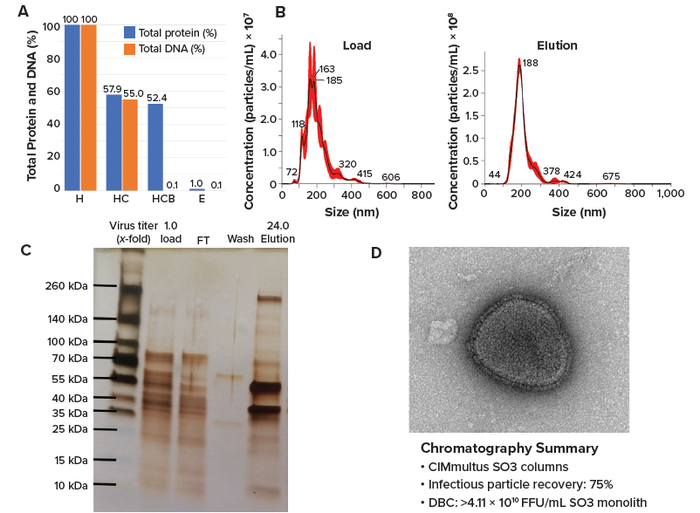
Figure 3: Analytics of NDV purification using a 1-mL CIMmultus SO3 monolith; (a) shows depletion of protein and DNA content from harvest (H) to elution. Panel (b) depicts particle-size distribution at loading and elution, (c) presents results from sodium dodecyl sulfate–polyacrylamide gel electrophoresis (SDS-PAGE), and (d) shows a transmission electron microscopy (TEM) scan of the elution fraction (e). (DBC = dynamic binding capacity, FT = flowthrough, HC = clarified harvest, HCB = Benzonase-treated clarified harvest).
Results from sodium dodecyl sulfate–polyacrylamide gel electrophoresis (SDS-PAGE) showed that most protein impurities from the load material appeared in the FT fraction. Virus eluted at high concentrations, generating intense bands representing viral proteins (16). Protein impurity levels were reduced in the eluted fraction, and confirmation of protein-impurity removal was obtained using a Bradford assay (Figure 3a and 3c). We determined the dynamic binding capacity (DBC) to be >4.11 × 1010 FFU/mL of SO3 monolith. Transmission electron microscopy (TEM) confirmed the presence of intact NDV particles in the eluate (Figure 3d).
Enabling Vaccine Manufacturing
Besides current applications in veterinary vaccines, NDV has considerable potential as a vector for treatment of infectious diseases and cancers in humans. Introducing CCX.E10 suspension cells into NDV production enables drug manufacturers to operate independently of SPF chicken-egg supply, and the line contains no egg-derived allergens. Its upstream process is well controlled and consistently generates high viral titers. Downstream purification can be performed in one chromatography step, with high yield and purity. The downstream process can provide a drug substance within a day.
The production and purification processes outlined above represent a robust manufacturing tool. The methodology described both supports optimization of NDV processes and enables biopharmaceutical companies to develop new NDV products.
References
1 Bukreyev A, Collins P. Newcastle Disease Virus as a Vaccine Vector for Humans. Curr. Opin. Mol. Ther. 10(1) 2008: 46–55.
2 Park JG, et al. Immunogenicity and Protective Efficacy of an Intranasal Live–Attenuated Vaccine Against SARS-CoV-2. iScience, 24 September 2021: 102941; https://doi.org/10.1016/j.isci.2021.102941.
3 de Swart RL, et al. Intranasal Administration of a Live–Attenuated Recombinant Newcastle Disease Virus Expressing the SARS-CoV-2 Spike Protein Induces High Neutralizing Antibody Levels and Protects from Experimental Challenge Infection in Hamsters. Vaccines 40(33) 2022: 4676-4681; https://doi.org/10.1016/j.vaccine.2022.07.005.
4 Ponce-de-León S, et al. Safety and Immunogenicity of a Live Recombinant Newcastle 1 Disease Virus–Based COVID-19 Vaccine (Patria) Administered via the Intramuscular or Intranasal Route: Interim Results of a Non-Randomized Open Label Phase I Trial in Mexico. medRxiv, 9 February 2022 (preprint); https://doi.org/10.1101/2022.02.08.22270676.
5 Yoshida A, et al. Novel Avian Paramyxovirus-Based Vaccine Vectors Expressing the Ebola Virus Glycoprotein Elicit Mucosal and Humoral Immune Responses in Guinea Pigs. Sci. Rep. 9, 2019: 5520; https://doi.org/10.1038/s41598-019-42004-4.
6 Kim SH, et al. A Novel Chimeric Newcastle Disease Virus Vectored Vaccine Against Highly Pathogenic Avian Influenza Virus. Virology 503, 2017: 31–36; https://doi.org/10.1016/j.virol.2017.01.006.
7 Wang J, et al. Newcastle Disease Virus-Vectored West Nile Fever Vaccine Is Immunogenic in Mammals and Poultry. Virol. J. 13, 2016: 109; https://doi.org/10.1186/s12985-016-0568-5.
8 Manoharan V, et al. Modified Newcastle Disease Virus as an Improved Vaccine Vector Against Simian Immunodeficiency Virus. Sci. Rep. 8, 2018: 8952; https://doi.org/10.1038/s41598-018-27433-x.
9 Liu T, et al. Optimization of Oncolytic Effect of Newcastle Disease Virus Clone 30 By Selecting Sensitive Tumor Host and Constructing More Oncolytic Viruses. Gene Ther. 28, 2021: 697–717; https://doi.org/10.1038/s41434-020-0145-9.
10 Song H, et al. Application of Newcastle Disease Virus in the Treatment of Colorectal Cancer. World J. Clin. Cases 7(16) 2019: 2143–2154; https://doi.org/10.12998/wjcc.v7.i16.2143.
11 Tajnik Sbaizero M, et al. Intensification of Influenza Virus Purification. BioProcess Int. 16(10) 2018: 32–38; https://bioprocessintl.com/downstream-processing/chromatography/intensification-of-influenza-virus-purification-from-clarified-harvest-to-formulated-product-in-a-single-shift.
12 WHO TRS 978. Recommendations for the Evaluation of Animal Cell Cultures as Substrates for the Manufacture of Biological Medicinal Products and for the Characterization of Cell Banks: Annex 3. World Health Organization: Geneva, Switzerland, 22 May 2013; https://www.who.int/publications/m/item/animal-cell-culture-trs-no-978-annex3.
13 Reiter M, et al. Avian Suspension Culture Cell Lines for Production. Industrial Scale Suspension Culture of Living Cells. Meyer H-P, Schmidhalter DR, Eds. Wiley: Hoboken, NJ, 2014: 503–554; https://doi.org/10.1002/9783527683321.ch11.
14 The Novel Suspension Quail Cell Line CCX.E10 Is an Ideal Cell Substrate for NDV Virus and Vector Production. Nuvonis: Vienna, Austria, 2022; https://nuvonis.com/ideal-substrate-for-virus-and-vector-production.
15 Rush BS, et al. In Vitro Infectivity of Oncolytic Newcastle Disease Virus: Correlation Between Plaque and Fluorescent Focus Assays. J. Virol. Meth. 251, 2018: 69–74; https://doi.org/10.1016/j.jviromet.2017.09.029.
16 Sun W, et al. A Newcastle Disease Virus Expressing a Stabilized Spike Protein of SARS-CoV-2 Induces Protective Immune Response. Nature Comm. 12, 2021: 6197; https://doi.org/10.1038/s41467-021-26499-y.
Hana Jug is head of product-application area for enveloped viruses, Natalija Hosta is associate scientist in downstream process development for enveloped viruses, Mojca Tajnik Sbaizero is head of the product-application area, and Aleš Štrancar is managing director, all at Sartorius BIA Separations, Mirce 21, SI-5270 Ajdovščina, Slovenia. Daniela Brodesser, Bianca Tisch, and Theresa Heger are laboratory scientists; Markus Wolschek is cofounder and chief scientific officer; Joachim Seipelt is head of business development; and corresponding author Manfred Reiter is cofounder and managing director of Nuvonis, Mariahilfer Str. 101/1/21, 1060 Vienna, Austria; 43-670-2024606; [email protected].
You May Also Like



.jpg?width=700&auto=webp&quality=80&disable=upscale)


