Voices of Biotech
Podcast: MilliporeSigma says education vital to creating unbreakable chain for sustainability
MilliporeSigma discusses the importance of people, education, and the benefits of embracing discomfort to bolster sustainability efforts.
December 1, 2009
Some of the numerous feeding strategies are more appropriate than others for certain types of cell culture production systems. Once a nutrient supplement has been identified as described in Part 1 of this three-part review (1), a supplementation strategy must be chosen. Supplementing at too great a rate may expose log-phase cells to stresses such as increased osmolality and lactate levels that would inhibit biomass expansion. But inadequate supplementation can lead to early apoptosis through rapid depletion of selected important components. For commercially available supplements, guidelines often suggest specific protocols that will usually yield good results and can be further optimized by subsequent experimentation.
Additional culture-specific methods are available to laboratories with access to monitoring equipment. As mentioned in Part 1, monitoring one component is an excellent way of approaching relative nutrient homeostasis in the bioreactor, especially with stoichiometrically balanced nutrient supplements (1). They may be added semicontinuously (e.g., daily or twice daily), with each addition providing enough supplement to cover its expected consumption until the next addition. Supplements also can be added continually to match predicted consumption, but this approach is more generally applicable at the research scale than in commercial production.
PRODUCT FOCUS: ALL PROTEINS
PROCESS FOCUS: PRODUCTION
WHO SHOULD READ: MANUFACTURING AND PROCESS DEVELOPMENT
KEYWORDS: CULTURE MEDIA, FED-BATCH, PERFUSION, MEDIA SUPPLEMENTS, PROCESS CONTROL, AND DESIGN OF EXPERIMENTS
LEVEL: INTERMEDIATE
Supplementation of cell cultures generally refers to adding nutrients that replace those consumed, but another type of supplement can be considered. Cell metabolic pathways are important to optimizing protein expression because of the dichotomous pathway of biomass expansion and product synthesis. Cells that are expanding rapidly may not have the highest specific productivity (output per cell), but once maximum cell density is reached, specific productivity can peak and be held at high levels for extended periods of time. Nutrient supplements can be designed to promote both phases. So nutritional supplements (those containing major nutrients such as glucose and specific amino acids that are consumed during the expansion phase) represent one type of supplement. And metabolic supplements (those containing molecules to guide cells toward maximum productivity) represent the other type. Timing their addition may promote superior productivity.
A number of molecules have been identified as helpful in the production phase of cell culture. When glucose is replaced by a more slowly metabolized hexose such as galactose or mannose, a slower metabolism results with correspondingly lowered lactic acid production. At the same time, inducers of protein synthesis such as butyrate and pentanoic acid can be used for switching cell machinery from further replication at the plateau phase into protein expression. Numerous small molecules are being researched as protein synthesis stimulators in high-throughput systems.

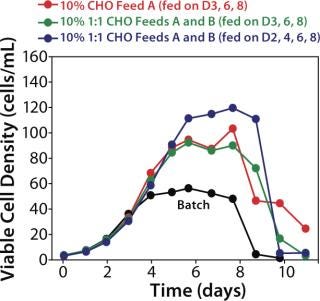
Nutrient Supplementation of Bioreactors
Bioreactor supplementation choices are made based on the timing and quantity of supplement to add to a culture. If a balanced, clone-specific nutrient supplement is used, one measurable component can determine the choice. With a low-glucose set point, Fike et al. (2) used computer-controlled peristaltic pumping of a stoichiometrically balanced supplement in a continuous fed-batch process to maintain a homogeneous nutrient environment that yielded a fourfold productivity increase.
Another relatively common method of determining when to add supplement is by monitoring OUR, the oxygen uptake rate (3). Tracking OUR to maintain nutrient levels provides an aerobic culture metabolism for maximum energy efficiency while minimizing lactate and ammonia production (3). This approach works whether supplementing just glucose and/or glutamine or a more complex supplement. Supplement is added based on oxygen consumption to control glucose and glutamine at low levels without depletion or overabundance. OUR-based supplementation can increase cell densities and improve productivity. With proper control, metabolism shifting toward increased lactate production during the growth phase may be counterbalanced by lactate consumption during the production phase (4).
Other methods of supplementation involve monitoring parameters such as pH, lactic acid, glutamine, dissolved oxygen (DO) levels, or glucose (3). Even turbidity can be monitored for supplementation purposes. Another approach is to add supplement proportional to the amount of base addition used to control pH. As a culture expands in cell number and consumes nutrients, it usually produces lactate, which reduces pH (kept in line by base addition and thus the cue for adding more supplement).
In another example of fed-batch process control, Sauer et al. used off-line measurement of glucose to guide supplementation of a concentrated protein-free supplement that was scalable from 15 L to 750 L, with a 7.6-fold improvement of monoclonal antibody (MAb) yield (5). Their approach was to seek a rapid, generic, fed-batch process using a commercially available “low-content” base medium and feeding the culture with a “rich” supplement. Somewhat analogous to this, Xie et al. specifically designed a base medium to reduce lactate and ammonia, then supplemented their culture with nutrients stoichiometrically shown to be depleting, allowing supplementation only as needed (6). Nutrients were kept from becoming depleted, lactate and ammonia levels were significantly lower, and cell density and productivity were increased by fivefold and 10-fold respectively.
Spens et al. (7) used a dual approach to derive a supplement that boosted productivity >11-fold. First, they performed depletion studies on bioreactor cultures to identify those macronutrients that could be quantitated easily. Nonmeasurable compounds were then identified by separate shake cultures of various supplement categories in a design of experiments (DoE) study. Combining data yielded a superior supplement that included lipids as an important feed component.
Fractional factorial design is another useful approach when it comes to assessing micronutrients for supplementation — e.g., trace elements, growth factors, and insulin — that may not be readily quantitated or that can interact with other supplement component(s). Sandadi et al. showed identification and quantitative modification of important components with interdependencies within a supplement (8).
Although most current supplementation regimens are of the nutrient-only type, biphasic approaches may become popular if they can enhance productivity. Success would require some knowledge of cell growth kinetics. Throughout the exponential phase, only nutrient supplements should be used to stimulate maximal cell mass expansion. Only as a culture has entered the plateau phase (but before a significant drop-off in viability) should a metabolic supplement such as butyrate be used.
Timing for addition of a metabolic inducer type of supplement (to immediately inhibit cell growth in favor of improved protein production) may need additional clarity. Sitton et al. (9) showed with CHO culture that monitoring gross viability or total cell count is not reliable for identifying the end of expansion and beginning of the culture’s stationary production phase. The team noted significant variation despite control of bulk culture parameters. They found that a nonviable subpopulation of cells (identified by automated flow cytometry) precisely identified the end of exponential proliferation, signaling the start of production feeding. The result was significantly improved total bioreactor cell count available for the production phase.
One other aspect of supplementation involves the potential need to replace nonconsumed components. In fed-batch cultures, up to 40–50% supplementation may be optimal. Nonconsumed chemicals thus may end up at half the concentration of that in the original medium, potentially dropping them to suboptimal levels. It may be advisable to add back some important components to yield a 1× “original” concentration. This may be especially important for vitamins, trace elements, and certain salts such as phosphates that have been shown to enhance cell product synthesis (10).
Supplementation Rates: A disadvantage of perfusion methods is product dilution resulting in high purification volumes. One solution may be combining multiple rounds of perfusion rate reduction with a more concentrated basal perfusion medium (11). Greater nutrient concentrations in the medium should allow perfusion rates to be lowered. Presumably this could provide much better productivities than fed-batch culture because some wastes are reduced even with a lowered perfusion rate. The challenge would be to choose a rate yielding the highest product concentration that also keeps toxic molecules at relatively low levels. Another limitation would be finite nutrient solubility in the basal medium — and the potential for adding grossly elevated levels of nutrients early in such a culture.
Slow feeding of concentrates with a fed-batch process based on known nutrient requirements usually improves productivity. Product synthetic pathways, however, may require minimal levels of sensitive or critical components that could easily be left out or added at too low a rate if consumption rates are unknown, jeopardizing product formation. Senger et al. compared a fixed, rapid feeding rate with a metabolically determined one and showed improved productivity using the former, pointing out that it may be better to err on the side of higher supplementation rates to cover unquantified cell needs (12). Using similar reasoning, Takagi et al. supplemented basal medium with fivefold amino acids and vitamins to increase volumetric yield of tissue plasminogen activator (tPA) by 3.6-fold, which the team considered to be critical to an economically viable production process (13).
In addition to stable production platforms, transient gene expression (TGE) is also benefited by nutrient supplementation (14,15). Sun et al. improved HEK 293 production of green fluorescent protein (GFP) and erythropoeitin (EPO) by supplementing with a 5× amino-acid concentrate based on DMEM/F12 in a fed-batch culture (15). Yet another approach was taken by Dempsey et al. using GS NSO cells adapted to grow in a serum-free medium without glutamine. The team used several rounds of culture supplementation followed by analytical spent-medium analysis to sequentially develop a nutrient supplement. In the final version, it showed depletion of no basic components and resulted in a final 10-fold improvement of product expression (16).
In an attempt to standardize methods of culture nutrient supplementation, Zeng et al. studied numerous stoichiometric ratios and related various cell culture parameters together (e.g., ammonium yield from glutamine or lactate from glucose) to determine which were most significant to cell productivity (17). They also presented an equation specifying oxygen consumption. The observed ratios were relatively constant and cell-line independent, making it possible to use them in controlling difficult-to-determine components and develop relatively fast feeding strategies for a number of cell lines and media. The team found that amino acid metabolism is a major consumer of oxygen and that using an OUR/glucose ratio is an excellent way to monitor and control culture supplementation rates.
Although nutrient supplementation is often thought to be important mainly as a culture reaches plateau and available nutrients are lowered or depleted, evidence suggests that considering a day-0 feed may prove beneficial. Fassnacht et al. showed that supplementation at the start of a culture led to decreased lactate and increased membrane stability during the death phase and increased MAb production. Addition at the beginning of the culture promoted beneficial metabolic pathways from initiation (18). Although components for a day-0 feed could be added to the basal medium itself for the same effect, adding them separately kept the basal medium “unadulterated” for use in a range of different clone culture options.
Metabolic Shift
Many articles use the term metabolic shift to imply adapting cells to a low-glucose, low-glutamine environment that reduces ammonia and lactate for improved protein productivity (20,21,22). But metabolic shifts in cell populations can be obtained through a number of chemical and physical approaches.
Nutrient Induced: Two key goals of nutrient supplementation are to maintain cells in the log growth phase to yield maximum biomass and to extend the productivity of that cell mass. Literature is replete with successful approaches for different cell culture production systems and related types of nutrient supplements. As an example, it has been observed for some cultures that after rising to relatively high levels, lactate will decrease to low levels during extended culture as it is consumed. Lactate rise relates to glucose consumption during early culture; as glucose becomes depleted (as in batch cultures), cells are forced to use lactate as an energy source (19). High lactate consumption rates can be observed even after high initial glucose concentrations of ≥15 g/L.
One approach to extending culture productivity involves a fairly complicated process known as metabolic shift. Historically it’s been observed that cell numbers and total productivity sometimes could be dramatically increased with lowered culture concentrations of waste products such as lactic acid and ammonia. As researchers subjected cells to reduced glucose and glutamine levels to force that change, they noticed that cells became much more efficient in their use of those nutrients over a number of generations (20,21). Cells could expand nicely in media containing a fraction of both glucose and glutamine concentrations, resulting in lowered levels of lactic acid and ammonia and presumably leading to increased cell numbers and significantly higher productivity levels (22). Cells were “adapting” from a glycolytic (fermentative) state to a more oxidative (aerobic) state.
With that approach, Lee et al. used a low-glutamine fed-batch feedback control-loop process in attempting to control ammonia and lactate with 293-HEK culture for adenovirus production (23). Controlling glutamine levels at 0.1 mM — with no other modifications — improved cell density and gave a 10-fold improvement in virus titer. And Li et al. controlled glucose at 0.3 mM and glutamine at 0.5 mM, which related to ammonia and lactate levels decreasing by 74% and 63% respectively (24). Their cultures extended from eight to 14 days, with a 1.7-fold increase in MAb titers. The team found that if glucose and glutamine levels became too low, other amino acids would be used instead.
In fact, the rate of glutamine feeding can determine the rate other amino acids are consumed (25), making it possible to begin planning supplements around those chemicals that do not lead to significant toxic waste product generation. Amino acid metabolism plays an important role in reducing lactate production (26). Under different growth conditions, cells can metabolize nutrients very differently while growing at the same rate. Glutamate can be used instead of glutamine as an energy source — especially but not exclusively in Lonza’s glutamine synthetase (GS) expression system — and can lower not only ammonia, but also cell consumption of lactate (27).
Bear in mind one caveat to a low-glucose, low-glutamine feeding strategy: Glucose or glutamine starvation can increase caspases and presumably lead to apoptotic pathways (28). So a very narrow concentration range must be maintained, with the ability to react rapidly to sudden excessively reduced glucose/glutamine levels. If for some reason nutrient feeding is halted or too slow, a cell culture can degrade rapidly. Hwang et al. showed that nutrient starvation during culture can cause both autophagy (enzymatic self-digestion) and apoptosis (programmed cell death) identified by different markers (29).
One other aspect of inducing metabolic shift should be considered: When cells are subjected to the long metabolically stressful process of initiating metabolic shift, repeated testing is needed to show that a product is glycolytically the same as before the change. In addition to metabolic shift methodology, any type of physical or chemical manipulation that alters cell metabolic state should be considered as initiating a metabolic shift. So several other mechanisms also belong in this category.
Decoupling growth from production is another approach to maintaining cells in the production phase. In one example involving CHO-based production of tPA, glucose was replaced by galactose (a slowly metabolized carbon source) to change cell behavior upon supplementation (30). Cell expansion essentially stopped, but the cell mass viability was maintained much longer in an oxidative (aerobic) instead of a fermentative state, resulting in significantly less generation of lactic acid. In another CHO feeding study, replacing glutamine by glutamate resulted in significantly less production of waste products (31). Glucose/glutamate ratios were studied, and it was feasible to increase cell growth efficiency by controlling glucose levels. But holding them too low negatively affected specific productivity, leading the authors to suggest that a combined use of alternative molecules with known controlled feed rates could maximize protein productivity.
In another CHO-based tPA system, glucose and glutamine were substituted by the slowly metabolized chemicals galactose and glutamate respectively (32). Amino acid cell culture use dictated formulation of both the base feed media. In addition, base medium salt was reduced so nutrient feeding could be continued beyond the point at which standard medium osmolality would limit supplementation. Waste products were lowered and cell viability and productivity increased as both glutamate and lactate were consumed for energy.
Culture “Stress” As a Potential Positive-Productivity Factor: In a kind of diabolical approach based on observations, applying “stressful” conditions will increase productivity in a number of instances. Such an approach could be considered after cultures have reached optimal cell expansion (33). Yoon et al. showed a 5× increase in CHO-cell erythropoietin production by combining osmotic, low culture temperature, and butyrate-induced growth inhibition techniques (34). This affected product glycosylation, leading to altered isoforms, which emphasized the importance of reassessing product quality after using alternate supplementation strategies. Further substantiating that are the results of Sandadi et al., who showed that high osmolality in the production phase improved protein yield as long as it was kept low enough during growth to obtain the highest possible expansion cell mass (35).
Culture pH Affects Cell Protein Production: In a GS-NSO recombinant MAb system, Osman et al. studied pH shifts from 7.3 to a range of values (36). The team showed that abrupt pH shift from pH 7.3 to pH >8.0 or <7.0 caused rapid apoptotic cell death (within 10 minutes). After the pH shift, maximum growth rate was observed in the pH range of 7.3–7.5, but maximum volumetric antibody titers were seen at pH 7.0. No alteration of the glycoprotein structure was seen at any pH range tested. After returning to the original pH, cell growth characteristics returned to normal. This study supports use of adequate mixing to equilibrate culture pH rapidly after base or nonneutral supplement addition to ensure consistent lot-to-lot performance.
A mathematical model is published for attaining pH control in bioreactors through describing the relationship between pCO2 (carbon dioxide partial pressure), pH, lactate, and base addition in a bicarbonate-buffered cell culture process (37). This model also considers use of carbonate for pH control as well as buffering by amino acids. The model has been shown relevant at T-flask up to 10,000-L fed-batch bioreactor sizes, which is especially important because pCO2 can be a problem at scale.
Temperature Reduction: A well-known observation is that lowering the temperature of a cell culture can promote a change from growth to protein production. Typically a culture is maintained at 37°C for maximal cell expansion. Once that has been achieved, the temperature is reduced several degrees depending upon the cell type. Fox et al. showed in a CHO–interferon system that expanding cells at 37°C and then lowering temperature to 32°C increased their yield by 90% over maintaining temperature at 37°C (38). The lower temperature maintained those cells viable in their productive phase longer — which presumably caused the improvement. The authors presented a modeling system for temperature shift that could be used for other cell lines.
In a CHO-based granulocyte-macrophage colony stimulating factor production system, a 2.1-fold improvement in specific productivity was seen with reduced growth rate, reduced lactate, and a prolonged stationary (production) phase (39). Interestingly, productivity improvements with lowering temperature may be not only cell line specific, but also product specific. Yoon et al. presented convincing evidence that cell production of CHO-based anti-4-1BB did not improve with temperature reduction (40). Longer cell viability was countered by suppressed growth and cell cycle arrest. Molecular analysis also supported the team’s results. Production of EPO in the same cell line, however, did show significant improvements through temperature reduction. So this technique for increasing production may be an option, but it’s not a sure thing.
Temperature-shift strategies may also be relevant to perfusion cultures. Perfusion typically produces high cell yields because of efficient removal of wastes and delivery of nutrients. Even though that is combined with the advantages of removing product to refrigerated storage to reduce protease action, titers can still be low because of product dilution. To improve perfusion titers, Yoon et al. used temperature shift for follicle-stimulating hormone (FSH) production (41). They grew cells up to 3.2 × 107/mL at 37°C, then shifted down to 28°C for a 13-fold productivity increase over leaving the culture at 37°C.
Carbon Dioxide: Although not usually considered as such, pCO2 should be thought of as toxic just like ammonium and lactate. For high-density/large-volume bioreactors, in which aeration may be insufficient to flush out high levels, pCO2 levels can negatively affect cell growth, metabolism, productivity, and product glycosylation (42). Possibilities for pCO2 reduction include reducing or eliminating sodium bicarbonate as a buffer, either replacing it with organic buffers such as MOPS-histidine or making pH adjustments with sodium carbonate concentrates instead (43). Doing so, Goudar et al. obtained a 63–70% reduction in pCO2, a 68–123% increase in growth rate, and a 58–92% improvement in specific productivity. Takuma et al. reported a similarly negative affect on growth with low glucose and higher pCO2 in a CHO perfusion bioreactor system (42). Previously those components were thought to be acting independently. In another study, Hu et al. reported significantly increased cell inhibition caused by increased CO2 levels at high osmolality, which occurs as cultures are supplemented (44).
Multiple Toxins: Although most research looks at individual toxic components, Xing et al. provided a guideline for assessing the real-world condition of multiple toxins acting on a culture at the same time (45). Using multivariate analysis, repression of threshold values for lactate, ammonia, osmolality, and pCO2 acting together are postulated and then tested to create a system that could be used for a range of production cell types.
Bioreactor Evolution: Another approach for optimizing and extending culture life span through a metabolic shift involves bioreactor dynamics. Termed bioreactor evolution, this process uses the bioreactor to “adapt” cells, analogous to what is done in flasks. Using host DG44 CHO cells, a doubling of peak cell density was observed after several bioreactor runs using the same cells (46). With a recombinant production CHO line, researchers obtained a 36% improvement in titer with no differences in product quality. Cells presumably became adapted to the bioreactor stress conditions through a type of natural selection. Although this process may be worth assessing — as opposed to initiating cultures with new freezes into flasks each time — other considerations related to bioreactor adaptation (e.g., maintaining sterility and genetic drift) need to be assessed as well.
Protein Production Enhancers
A number of small-molecule protein production enhancers work through various metabolic pathways to inhibit cell expansion, thus switching cells into a production phase. These molecules tend to be somewhat cytotoxic, supposedly working through stimulation of apoptotic pathways, so their use must be assessed for each cell line. But these enhancers have been known to double productivity.
Several mechanisms of butyrate action have been suggested such as enhanced gene transcription through gene accessibility stimulation (47). In one study, sodium butyrate and sodium propionate (an alkanoic acid) enhanced MAb production sixfold (48). Whereas butyrate showed toxicity, no cell inhibition was seen with propionate. Similar results were reported comparing pentanoic acid (a carboxylic acid) with butyrate (49).
Another study tested valproic acid (which is both FDA approved and much less expensive than butyrate) as an alternative to butyrate (50). Productivity improvements were observed. Seeking new protein-inducing compounds, Allen et al. used a high-throughput model for screening chemical libraries to identify several basic types of molecules as potential possibilities (51). Cell transcriptional activity helped in the team’s search and points to use of molecular approaches in future efforts.
In a different mode, adenosine 5′-monophosphate (AMP) significantly increased hepatitis B surface antigen production in CHO cells but dramatically reduced cell number once added to a culture (52). Extracellular nucleotides were shown to enhance protein production because intracellular adenosine 5′-triphosphate (ATP) pools increased with AMP addition. Yet another study showed rapamycin (a G1-phase arresting agent that slows cell cycle progression to delay apoptosis) to reduce hybridoma cell death and enhance MAb production twofold in fed-batch cultures (53). Dimethyl sulfoxide (DMSO) has also been advocated as a protein-inducing enhancer. Ling et al. showed that 0.2% DMSO added to hybridoma cultures at maximum cell density doubled MAb production with no negative effect on bioactivity and glycosylation (54).
The Next Step
As reviewed here, different nutrient supplements and feeding strategies can improve cell performance. Once basic cell culture parameters have been optimized, nutrient supplementation can usually result in a doubling or greater of cell productivity. The conclusion of this three-part review will examine scale-up and scale-down strategies for rapid nutrient supplement prototyping.
1.) Fike, R. 2009. Nutrient Supplementation Strategies for Biopharmaceutical Production, Part 1: Identifying a Nutrient Supplementation Formulation. BioProcess Int. 7:44-51.
2.) Fike, R. 1993. Feeding Strategies for Enhanced Hybridoma Productivity: Automated Concentrate Supplementation. BioPharm 6:49-54.
3.) Wlaschin, K, and W-S. Hu. 2006. Fedbatch Culture and Dynamic Nutrient Feeding. Adv. Biochem. Engin./Biotechnol. 101:43-74.
4.) Zhou, W.. 1997. Fed-Batch Culture of Recombinant NS0 Myeloma Cells with High Monoclonal Antibody Production. Biotechnol. Bioeng. 55:783-792.
5.) Sauer, P. 2000. A High-Yielding, Generic Fed-Batch Cell Culture Process for Production of Recombinant Antibodies. Biotechnol. Bioeng. 67:585-597.
6.) Xie, L, and D. Wang. 2006. Fed-Batch Cultivation of Animal Cells Using Different Medium Design Concepts and Feeding Strategies. Biotechnol. Bioeng. 95:270-284.
7.) Spens, E, and L. Haggstrom. 2007. Defined Protein and Animal Component-Free NS0 Fed-Batch Culture. Biotechnol. Bioeng. 98:1183-1194.
8.) Sandadi, S. 2006. Application of Fractional Factorial Designs to Screen Active Factors for Antibody Production By Chinese Hamster Ovary Cells. Biotechnol. Prog. 22:595-600.
9.) Sitton, G, and F. Srienc. 2008. Mammalian Cell Culture Scale-Up and Fed-Batch Control Using Automated Flow Cytometry. J. Biotechnol. 135:174-180.
10.) deZengotita, V. 2000. Phosphate Feeding Improves High-Cell-Concentration NSO Myeloma Culture Performance for Monoclonal Antibody Production. Biotechnol. Bioeng. 69:566-576.
11.) Konstantinov, K. 2006. The “Push-to-Low” Approach for Optimization of High-Density Perfusion Cultures of Animal Cells. Adv. Biochem. Engin./Biotechnol. 101:75-98.
12.) Senger, R, and M. Karim. 2007. Optimization of Fed-Batch Parameters and Harvest Time of CHO Cell Cultures for a Glycosylated Product with Multiple Mechanisms of Inactivation. Biotechnol. Bioeng. 98:378-390.
13.) Takagi, M. 2001. Effects of High Concentrations of Energy Sources and Metabolites on Suspension Culture of Chinese Hamster Ovary Cells Producing Tissue Plasminogen Activator. J. Biosci. Bioeng. 91:515-521.
14.) Baldi, L. 2007. Recombinant Protein Production By Large-Scale Transient Gene Expression in Mammalian Cells: State of the Art and Future Perspectives. Biotechnol. Lett. 29:677-684.
15.) Sun, X. 2006. Enhancement of Transient Gene Expression By Fed-Batch Culture of HEK 293 EBNA1 Cells in Suspension. Biotechnol. Lett. 28:843-848.
16.) Dempsey, J. 2003. Improved Fermentation Processes for NSO Cell Lines Expressing Human Antibodies and Glutamine Synthetase. Biotechnol. Prog. 19:175-178.
17.) Zeng, A-P. 1998. Variation of Stoichiometric Ratios and Their Correlation for Monitoring and Control of Animal Cell Cultures. Biotechnol. Prog. 14:434-441.
18.) Fassnacht, D. 1997. Influence of Non-Essential Amino Acids on Apoptotic and Necrotic Death of Mouse Hybridoma Cells in Batch Cultures. Biotech. Lett. 19:35-38.
19.) Burky, J. 2007. Protein-Free Fed-Batch Culture of Non-GS NSO Cell Lines for Production of Recombinant Antibodies. Biotechnol. Bioeng. 96:281-293.
20.) Gambhir, A. 1999. Alteration of Cellular Metabolism By Consecutive Fed-Batch Cultures of Mammalian Cells. J. Biosci. Bioeng. 87:805-810.
21.) Wlaschin, K, and W-S. Hu. 2006. Fedbatch Culture and Dynamic Nutrient Feeding. Adv. Biochem. Engin./Biotechnol. 101:43-74.
22.) Maranga, L, and C. Goochee. 2006. Metabolism of PER.C6™ Cells Cultivated Under Fed-Batch Conditions at Low Glucose and Glutamine Levels. Biotechnol. Bioeng. 94:139-150.
23.) Lee, Y. 2003. Low-Glutamine Fed-Batch Cultures of 293-HEK Serum-Free Suspension Cells for Adenovirus Production. Biotechnol. Prog. 19:501-509.
24.) Li, L. 2005. Increasing the Culture Efficiency of Hybridoma Cells By the Use of Integrated Metabolic Control of Glucose and Glutamine at Low Levels. Biotechnol. Appl. Bioehem. 42:73-80.
25.) Vriezen, N. 1997. Effects of Glutamine Supply on Growth and Metabolism of Mammalian Cells in Chemostat Culture. Biotechnol. Bioeng. 54:272-286.
26.) Gambhir, A. 2003. Analysis of Cellular Metabolism of Hybridoma Cells at Distinct Physiological States. J. Biosci. Bioeng. 95:317-327.
27.) Kuwae, S. 2005. Development of a Fed-Batch Culture Process for Enhanced Production of Recombinant Human Antithrombin By Chinese Hamster Ovary Cells. J. Biosci. Bioeng. 100:502-510.
28.) Yeo, J. 2006. Glutamine or Glucose Starvation in Hybridoma Cultures Induces Death Receptor and Mitochondrial Apoptotic Pathways. Biotechnol. Lett. 28:1445-1452.
29.) Hwang, S, and G. Lee. 2008. Nutrient Deprivation Induces Autophagy as well as Apoptosis in Chinese Hamster Ovary Cell Culture. Biotechnol. Bioeng. 99:678-685.
30.) Altamirano, C. 2001. Decoupling Cell Growth and Product Formation in Chinese Hamster Ovary Cells Through Metabolic Control. Biotechnol. Bioeng. 76:351-360.
31.) Altamirano, C. 2001. Analysis of CHO Cells Metabolic Redistribution in a Glutamate-Based Defined Medium in Continuous Culture. Biotechnol. Prog. 17:1032-1041.
32.) Altamirano, C. 2004. Strategies for Fed-Batch Cultivation of t-PA Producing CHO Cells: Substitution of Glucose and Glutamine and Rational Design of Culture Medium. J. Biotechnol. 110:171-179.
33.) Miller, W. 1988. A Kinetic Analysis of Hybridoma Growth and Metabolism in Batch and Continuous Suspension Culture: Effect of Nutrient Concentration, Dilution Rate, and pH. Biotechnol. Bioeng. 32:947-965.
34.) Yoon, S. 2004. Effect of Simultaneous Application of Stressful Culture Conditions on Specific Productivity and Heterogeneity of Erythropoietin in Chinese Hamster Ovary Cells. Biotechnol. Prog. 20:1293-1296.
35.) Sandadi, S. 2005. Heuristic Optimization of Antibody Production By Chinese Hamster Ovary Cells. Biotechnol. Prog. 21:1537-1542.
36.) Osman, J. 2001. The Response of GS-NSO Myeloma Cells to pH Shifts and pH Perturbations. Biotechnol. Bioeng. 75:63-73.
37.) Gramer, M, and T. Ogorzalek. 2007. A Semi-Empirical Mathematical Model Useful for Describing the Relationship Between Carbon Dioxide, pH, Lactate and Base in a Bicarbonate-Buffered Cell-Culture Process. Biotechnol. Appl. Biochem. 47:197-204.
38.) Fox, S. 2004. Maximizing Interferon-γ Production By Chinese Hamster Ovary Cells Through Temperature Shift Optimization: Experimental and Modeling. Biotechnol. Bioeng. 85:177-184.
39.) Fogolin, M. 2004. Impact of Temperature Reduction and Expression of Yeast Pyruvate Carboxylase on hGM-CSF-Producing CHO Cells. J. Biotechnol. 109:179-191.
40.) Yoon, S. 2003. Effect of Low Culture Temperature on Specific Productivity and Transcription Level of Anti-4-1BB Antibody in Recombinant Chinese Hamster Ovary Cells. Biotechnol. Prog. 19:1383-1386.
41.) Yoon, S. 2007. Effect of Culture Temperature on Follicle-Stimulating Hormone Production By Chinese Hamster Ovary Cells in a Perfusion Bioreactor. Appl. Microbiol. Biotechnol. 76:83-89.
42.) Takuma, S. 2007. Dependence on Glucose Limitation of the pCO2 Influences on CHO Cell Growth, Metabolism and IgG Production. Biotechnol. Bioeng. 97:1479-1488.
43.) Goudar, C. 2007. Decreased pCO2 Accumulation By Eliminating Bicarbonate Addition to High Cell-Density Cultures. Biotechnol. Bioeng. 96:1107-1117.
44.) Hu, W-S, and J. Aunins. 1997. Large-Scale Mammalian Cell Culture. Curr. Opin. Biotechnol. 8:148-153.
45.) Xing, Z. 2008. Identifying Inhibitory Threshold Values of Repressing Metabolites in CHO Cell Culture Using Multivariate Analysis Methods. Biotechnol. Prog. 24:675-683.
46.) Prentice, H. 2007. Improving Performance of Mammalian Cells in Fed-Batch Processes Through “Bioreactor Evolution.”. Biotechnol. Prog. 23:458-464.
47.) Jiang, Z, and S. Sharfstein. 2008. Sodium Butyrate Stimulates Monoclonal Antibody Over-Expression in CHO Cells By Improving Gene Accessibility. Biotechnol. Bioeng. 100:189-194.
48.) Chun, B-H. 2003. Enhanced Production of Recombinant B-Domain Deleted Factor VIII from Chinese Hamster Ovary Cells By Propionic and Butyric Acids. Biotech. Lett. 25:315-319.
49.) Liu, C-H. 2001. Pentanoic Acid, a Novel Protein Synthesis Stimulant for Chinese Hamster Ovary (CHO) Cells. J. Biosci. Bioeng. 91:71-75.
50.) Backliwal, G. 2008. Valproic Acid: A Viable Alternative to Sodium Butyrate for Enhancing Protein Expression in Mammalian Cell Cultures. Biotechnol. Bioeng. 101:182-189.
51.) Allen, M. 2008. Identification of Novel Small Molecule Enhancers of Protein Production By Cultured Mammalian Cells. Biotechnol. Bioeng. 100:1193-1204.
52.) Luo, W. 2005. Enhancement of Hepatitis B Surface Antigen Production By Adenosine 5′-Monophosphate in Culture of Recombinant Chinese Hamster Ovary Cells. J. Bioscience Bioeng. 100:475-477.
53.) Balcarcel, R, and G. Stephanopoulos. 2001. Rapamycin Reduces Hybridoma Cell Death and Enhances Monoclonal Antibody Production. Biotechnol. Bioeng. 76:1-10.
54.) Ling, W. 2003. Improvement of Monoclonal Antibody Production in Hybridoma Cells by Dimethyl Sulfoxide. Biotechnol. Prog. 19:158-162.
You May Also Like