Voices of Biotech
Podcast: MilliporeSigma says education vital to creating unbreakable chain for sustainability
MilliporeSigma discusses the importance of people, education, and the benefits of embracing discomfort to bolster sustainability efforts.
November 9, 2010
Sponsored by BioScale
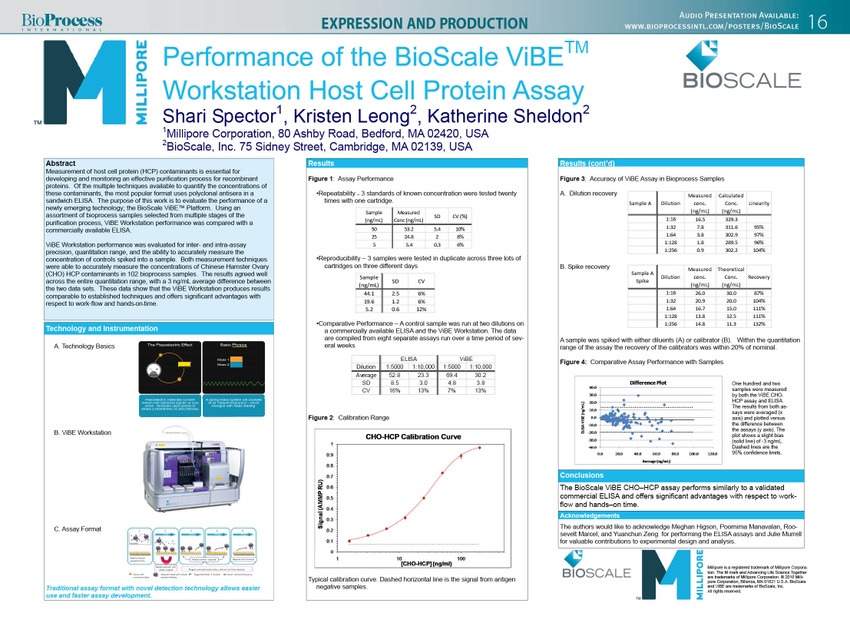
Measurement of host cell protein (HCP) contaminants is essential for developing and monitoring an effective purification process for recombinant proteins. Of the multiple techniques available to quantify the concentrations of these contaminants, the most popular format uses polyclonal antisera in a sandwich ELISA. The purpose of this work is to evaluate the performance of a newly emerging technology; the BioScale ViBE™ Platform. Using an assortment of bioprocess samples selected from multiple stages of the purification process, ViBE Workstation performance was compared with a commercially available ELISA.
ViBE Workstation performance was evaluated for inter- and intra-assay precision, quantitation range, and the ability to accurately measure the concentration of controls spiked into a sample. Both measurement techniques were able to accurately measure the concentrations of Chinese Hamster Ovary (CHO) HCP contaminants in 102 bioprocess samples. The results agreed well across the entire quantitation range, with a 3 ng/mL average difference between the two data sets. These data show that the ViBE Workstation produces results comparable to established techniques and offers significant advantages with respect to work-flow, flexibility and hands-on-time.
Contact
For questions about this poster, please contact:
Brett Masters, Chief Technical Officer, BioScale, Inc.
[email protected]
You May Also Like