Designing, Developing, and Delivering Our Clients’ Biopharmaceuticals July 2008
July 1, 2008

Eden Biodesign is one of Europe’s leading contract biomanufacturers. We are an expert provider of consultancy, biopharmaceutical process development, and CGMP manufacturing services. Our rarely found blend of experience and expertise, gained from providing our services to more than 100 clients worldwide, ensures that the clinical medicines we develop are regulatory compliant and optimally positioned for further clinical and commercial development. Our state-of-the-art facility is licensed to produce clinical products, which we believe is best in class for its scale of manufacture, whether for microbial or mammalian recombinant proteins, cell products, viral vaccines, or gene therapies.
Our primary goal is to be open and honest. We all know that the path to biopharmaceutical glory is not always smooth, but the truth is that we will never seek to hide anything from you. We will keep you informed of setbacks as well as breakthroughs. We don’t develop proprietary products, and although we provide access to the world’s leading production technologies, we don’t seek royalty payments. Providing a development and manufacturing service to our clients receives 100% of our attention and investment.
Eden Biodesign: Much More Than a CMO
Eden Biodesign is a fully integrated biopharmaceutical manufacturer and developer, offering expertise across the spectrum of development disciplines. For example, we are regularly hired to perform due diligence for clients in-licensing clinical biopharmaceuticals, to train biopharma manufacturing staff, and to prepare regulatory submissions, including BLAs. It is this rarely found blend of experience and expertise — coupled with our process and analytical development and manufacturing capabilities — that makes us a truly unique contract biopharmaceutical development and manufacturing partner.
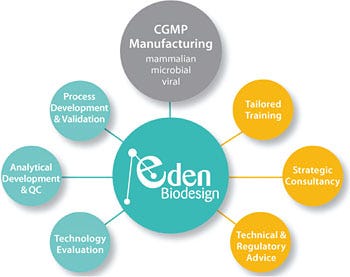
Our motto is “Begin with the end in mind,” a distillation of our experience gained over many years working with biotech and CMO clients. This has shown us that successful product development requires clear goals and good science from day one.
World Class Facilities Available to All
Our 4,100-m2 (43,000 ft2) facility based in Liverpool, UK, provides a range of dedicated and segregated suites for mammalian, microbial, and live-virus products — suitable for all stages of biopharmaceutical development. Our process development laboratories are scaled-down versions of our manufacturing suites with identical equipment platforms allowing rapid, low-risk technology transfer. Our manufacturing suites are highly flexible and can be rapidly configured to efficiently manufacture the most complex products. We are particularly proud of our commitment to quality, which has allowed us to sign up major pharma as well as leading biotech companies as clients for our CGMP services.
In November 2007, our Group established a US subsidiary — Eden Biodesign, Inc. — to make it easier for North American clients to access Eden’s proven expertise. Building on the strong track record and recent growth of our UK CGMP facility, it will offer a range of cell line/strain development and early process development services from its facility in Research Triangle Park, NC.
With satisfied clients in the United States, Europe, Asia, South America, Africa, and Australia, we provide a truly global service. To learn more about how we can assist your development programs and explore a new beginning, please contact us.
You May Also Like





