Speed to clinic testing — and then speed to market — are highly significant metrics for companies developing biopharmaceuticals. By increasing the pace of drug development, these companies can reduce costs, obtain revenues early, and establish commanding positions in the market relative to their competitors. High-throughput development tools have contributed much to the acceleration of drug development in recent years. Such technologies enable the testing of many process parameters in parallel. Combining them with multifactorial “design of experiment” (DoE) analysis leads to design of highly efficient and well-characterized bioprocesses.
One challenge that arises when companies adopt such an approach is that the manufacturing “bottleneck” shifts from process development groups to analytical testing laboratories. Small biotechnology companies often face a dilemma with respect to analysis of samples from early stage experiments: Either they must purchase expensive, sophisticated analytical technologies and employ the necessary human staff members to operate those effectively, or they must outsource the work to contract testing laboratories (CTOs) — often increasing the time it takes to obtain results.
So the biopharmaceutical industry is short of cost-effective analytical tools that enable rapid decision-making during product and process development. Companies need such tools to help them navigate their way to the clinic and beyond.
CASE STUDY: ADC CHARACTERIZATION
Antibody–drug conjugate (ADC) processes illustrate this dilemma. Process development scientists often want to screen a large number of conditions for the conjugation step to identify those that will deliver an optimum drug/antibody ratio (DAR). Development scientists can perform these experiments relatively quickly and at small scale to screen different methods of conjugation with a range of solvents. But then process development groups must ensure that the same conditions will yield conjugated products of adequate quality.
An effective ADC drug first must bind to its target receptor, then be internalized by the cell expressing that receptor to release cytotoxic payload molecules into the cytoplasm. End-point, cell-based assays will indicate whether those three steps happen effectively but often are accessible to small and medium-sized biotechnology companies only through contract research organizations (CROs). For that reason, many developers are choosing to assess the quality of their ADC products based on molecular analysis. Such testing can provide some information (e.g., the DAR), but it does not provide elucidation of functionality. So relying on molecular analysis alone can be misleading. Identifying conditions that give high DAR ratios to optimize toxophore loading is of no use if such high loading impairs an antibody from binding to its target.
Development engineers in the ADC industry need a method for analyzing ADC products during process development that allows for timely assessment of functionality without requiring costly cell-based analytical methods. Orla Protein Technologies Ltd. worked with Glythera Ltd. — an emerging biotechnology company focused on development of antibody-based treatments for cancer and other conditions — in creating one solution. They have developed a fusion protein consisting of a surface binding unit and the Her2 domain IV receptor for trastuzumab using Orla’s OrlaSURF protein engineering technology (1). That combination allows analysts to anchor the protein to a surface while controlling the structure and orientation of the target protein so to mimic its natural structural milieu.
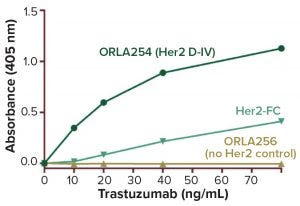
Figure 1: Enzyme-linked immunosorbent assay (ELISA) results show trastuzumab binding to ORLA254 engineered protein, control SBU (ORLA256), and native Her-2-Fc fusion produced in eukaryotic cells (from R&D Systems), with average data from three wells plotted. Display of domain IV on the OrlaSURF protein results in greater sensitivity of the assay than with adsorbed native Her-2 protein (limit of detection <10 ng/mL on OrlaSURF protein, 20 ng/mL for native protein). The lower detection limit means that much less precious ADC material needs to be used for analysis.
Because of the superior way the receptor presents itself, OrlaSURF technology provides for a more sensitive assay than absorbed native Her-2 protein can (Figure 1). This technique can be transferred to analytical biosensors for rapid, real-time, high-throughput analysis (2). The technique is applicable to not only ADCs, but also to a broad range of biologics including antibodies and vaccines.
ANALYTICAL METHODS FOR STRATEGIC ADVANTAGE?
It would be a mistake to underestimate the importance of having an optimized approach to analytics during development of biopharmaceutical products. Biopharmaceutical executives can make effective business decisions only if they have the correct information about molecules in development. This applies across the industry. For example, in the race to develop biosimilars, being able to assess comparability to a reference innovator molecule during development enables managers to gauge the likelihood of successful commercialization at each stage in the development cycle. Understanding that a biologic in development may not have the necessary quality attributes allows companies to make the difficult decision to terminate the project if necessary and focus their resources on alternative programs with greater chances of success.
Biopharmaceutical analytical technology is a field in which considerable innovation is both possible and highly desirable. Companies that can master the art of developing the best analytical approach for their programs have an opportunity to reduce costs while reaching clinical testing and then market approval more quickly than those who cannot. Armed with such competence, forward-looking biomanufacturers can achieve higher revenues and a greater share of the market than their less modernized competitors. Your analytical strategy could help your company achieve a competitive advantage.
REFERENCES
1 Shah DSH, Wernhart C, Athey D. Advanced Protein Engineering Enhances Biopharmaceutical Manufacturing and Analytics. BioProcess Int. 14(10) 2016: 46–52; www. bioprocessintl.com/2016/advanced-protein-engineeringenhances-biopharmaceutical-manufacturing-analytics.
2 Matthias KH, et al. A Protein-Engineering Approach to In-Process Assay Development During Ab-Drug Conjugate Manufacture: A Proof of Concept with Trastuzumab. (Poster) World ADC: San Diego, 2016; www.orlaproteins.com/wp-content/uploads/2016/01/ Poster_WADC-2016_ORLA.pdf.
When this article was first published (11 January 2017), Dr. Nick Hutchinson was a technical content marketing manager for Sartorius Stedim Biotech. Currently he is Saturn Business Unit Lead at FUJIFILM Diosynth Biotechnologies in Wilton, UK. This piece originally appeared online on the Next Generation Therapeutics community page. We are grateful to our KNect365 colleagues for permission to reprint.








