
HTTPS://WWW.ALAMY.COM
As the bioprocessing sector marches toward the future, digitalization stands at the heart of the Industry 4.0 vision of smart, self-organizing factories. Along with critical success factors such as infrastructure investment in cloud-based technologies, digitalization enables companies to turn data into intelligence. The realization of Industry 4.0 promises digitally integrated facilities with fully automated manufacturing, real-time traceability, standardized procedures, and agile processes (1, 2). Here, we present the results of a benchmarking survey that drew participation from leading biopharmaceutical companies. And we describe initiatives that UCL Biochemical Engineering is undertaking to address both the immediate and long-term digital skills needs of the sector.
Benchmarking DigitalMaturity and Skills Needs
Funded by UK Research and Innovation (UKRI) and hosted at University College London (UCL), the Future Targeted Healthcare Manufacturing (FTHM) Hub has held several events to debate the potential benefits of implementing digitalization strategies within the biotechnology sector, particularly for cell and gene therapies and protein biologics. The FTHM Hub assembles leading academics from UK institutions (e.g., UCL, Imperial, Manchester, Loughborough, and Warwick Universities) with a 45-member strong industrial consortium that actively engages in next-generation bioprocessing. Those companies include leading multinationals (e.g., AstraZeneca, GlaxoSmithKline (GSK), Lilly); small and mid-sized enterprises (MSEs); contract manufacturers and suppliers; industry associations; and UK translational research organizations (e.g., CPI and the Cell and Gene Therapy Catapult (CGTC)). In 2021, FTHM Hub participants answered survey questions to benchmark the digital maturity and skills needs of the bioprocessing sector (Figure 1). Demographically, 40% of respondents are biopharmaceutical developers or manufacturers, 30% are suppliers, and 30% are from trade associations, consultancies, and translational organizations.
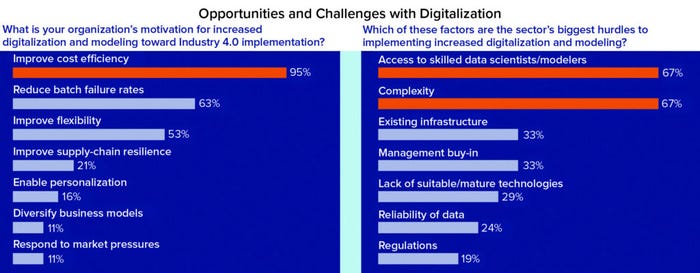
Figure 1a: Survey responses on benchmarking the digital maturity and skills needs of the bioprocess industry sector (AI = artificial intelligence, ML = machine learning, PCA = principal component analysis, PLS = partial least squares, SVM = support-vector machine, CART = classification and regression tree, NN = neural network).
Opportunities and Challenges with Implementation: The survey asked respondents to identify multiple benefits and impediments to increasing digitalization and modeling in the sector. Improving cost efficiency (95% of respondents) was identified as the top motivator for implementing digital strategies, followed by reducing batch-failure rates (63%), and improving flexibility (53%). The top two obstacles to implementing digital strategies were access to digital skills (67%) and perceived complexity (67%), followed by existing infrastructure (33%) and management buy-in (33%). These responses align with challenges identified by McKinsey & Company regarding digital implementation across the wider process industries, which cited cultural challenges, lack of understanding about digital trends, and lack of digital skills (3).
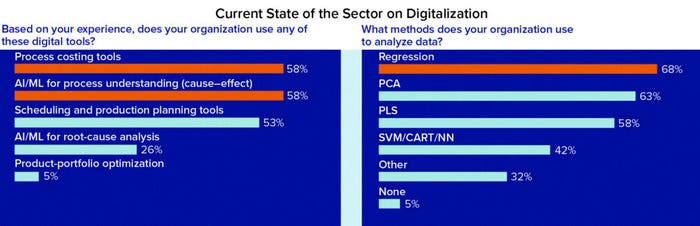
Figure 1b
Assessing the State of Digitalization in Bioprocessing: Survey respondents were asked how their organizations use artificial intelligence (AI) and machine learning (ML) technologies to analyze data. Their answers suggest that regression, partial least squares (PLS), and principal component analysis (PCA) techniques are the most popular applications (58–68%); fewer respondents use neural networks, classification and regression trees, and support-vector machines. Those results reflect ML adoption trends observed across the broader biopharmaceutical sector. Citation frequency in publications and patents show that over the past decade, PLS has been among the most popular techniques (4).
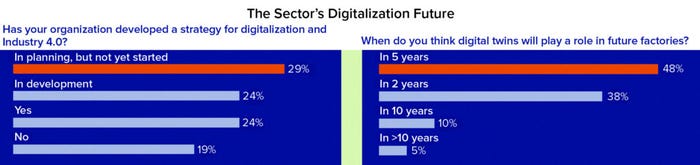
Figure 1c
Assessing Progress Toward Digitalization: We used this survey to assess how companies are moving toward implementing digitalization strategies. It revealed that 24% of respondents have started digital development, whereas another 29% are planning to do so. About half of respondents (~50%) predict that digital twins will play an increasing role in their facilities within five years, which suggests that upskilling is required for building the expertise and training to develop, implement, and maintain such models. However, it is worth noting that the term digital twin refers to different setups across different organizations that range from empirically based simulations to mechanistic models that adapt to real-time information and thus enable process control.
Below, we describe a holistic approach that UCL Biochemical Engineering takes to help repurpose existing skill sets as organizations prepare to adopt increasing levels of digitalization, in addition to equipping our next generation of graduates with the necessary digital skill set.
CPD Module on Developing Smart Digitalization Strategies
To achieve Industry 4.0 implementation, the bioprocessing sector needs to develop its workforce quickly with the skills necessary for using platforms outfitted with modern digital technologies. Modular courses offering continuous professional development (CPD) play a key role in upskilling current staff members.
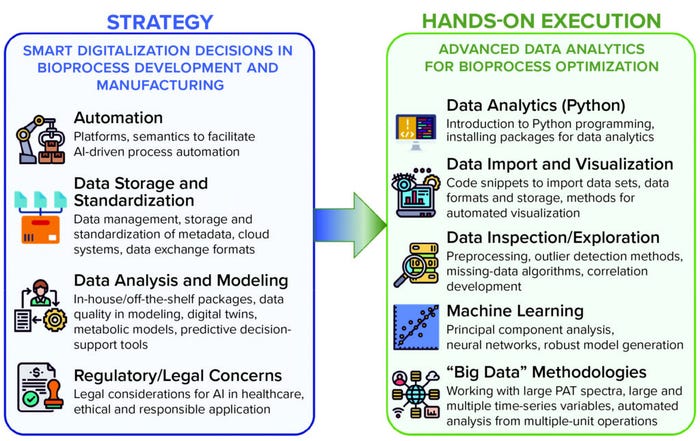
Figure 2: Relationship and content of the two new CPD MBI modules on digital skills at UCL; (left) Smart Digitalisation Decisions in Bioprocess Development and Manufacturing focuses on developing strategies to prepare a bioprocessing business to transition into a digital-compliant mode of operation; (right) Advanced Data Analytics for Bioprocess Optimisation is a hands-on module providing the necessary skill set to leverage useful and actionable information from complicated bioprocessing.
At a strategic level, bioindustry professionals such as group managers, team leaders, and consultants increasingly need to make informed decisions about adopting digital technologies in their operations at short notice. They also must offer advice on action plans to remain competitive within a rapidly evolving landscape. This, understandably, has proven to be a challenging path. The UCL Modular Training for the Bioprocess Industry (MBI) program (https://www.ucl.ac.uk/biochemical-engineering/study/industrial-training) has acknowledged the pressing need for familiarization with the breadth of concepts and technologies required for digital transformation. In 2022, we delivered a new MBI module on Smart Digitalisation Decisions in Bioprocess Development and Biomanufacturing, as shown in Figure 2.
The module aims to support the bioprocessing sector in making the best-fitting practical decisions to enable digital-readiness. It is designed primarily for bioprocess development decision-makers: project managers, chief scientific officers, investors and start-up owners, research managers, and consultants. The module supports developing strategies to identify critical choices and considerations for preparing biopharmaceutical businesses to transition into a digitally compliant mode of operation. It provides biobusiness decision-makers and managers with information on the benefits of digital readiness and the potential utility of digital technologies toward improving bioprocess development and operation.
We have structured that module under the following strategic themes: process automation, data storage and standardization for a digital future, data analysis pipelines and process models, and regulatory and legal concerns for artificial intelligence (AI) in biomanufacturing. Within each theme, we offer a platform for attendees to network with sector leaders and subject-matter experts from both industry and academia. We also enable them to hear from end users about first-hand experiences in adopting these technologies.
We believe that sharing good practice is of utmost importance. To enable that, we designed “Mind the Gap” sessions to which we invite end users and technology providers to offer their experiences with strategic partnerships to facilitate forms of digital transformation. The technology partners share personal success stories and discuss what they have learned about best practices. In 2022, such sessions enabled delegates to participate in discussions with developers and industry users of cloud-based automation platforms. Such upskilling of participants enables them to make informed decisions when developing their own strategies. We facilitate that further through discussion sessions for Implementation of Tailored Roadmaps Towards Digitalisation. That training module presents an excellent networking and benchmarking opportunity for participants as they advance their aspirations in the digital space within their respective corporations.
Through the module, we aim to equip decision-makers with an understanding of digital concepts that relate strongly to the bioprocess industries. However, that only partly addresses the immediate skills requirements of the sector. There is also a need to upskill practicing scientists and engineers with in-depth knowledge of digital techniques and tools that they can incorporate into their daily practices. We intend to achieve that through our new complementary module on advanced data analytics.
CPD Module on Hands-On Advanced Data Analytics Skills
Exponential growth of data within the biomanufacturing sector has created a major challenge over the past decade. Leveraging data for better-informed operational decisions requires a new breed of scientists and engineers with the necessary skill set and confidence to analyze their own data sets. Unfortunately, training has not kept pace with the rapid increase of complicated data sets, resulting in a major shortage of scientists and engineers with the ability to interpret them (2). To help address this skills gap, we started a short, modular, hands-on CPD module to introduce delegates to data science. The module teaches them how to apply the right algorithms and statistics to extract information from noisy, complex, and unstructured bioprocess data sets and leverage those insights for bioprocess optimization (Figure 2, right panel).
The module on Advanced Data Analytics for Bioprocess Optimisation helps professionals understand their data and better minimize repetitive visualization and analysis tasks while learning the fundamentals of data science. The module emphasizes the importance of shifting the sector away from its dependence on spreadsheets, which both limit opportunities for automated workflows and store data inconsistently. This module uses Python, a powerful and flexible open-source programming language that is becoming widely used across the industry because developers have designed abundant libraries for data manipulation and analysis.
The CPD module was launched in 2021. It provides delegates with specific skills required to use Python and analyze real-world data sets. It also teaches them how to use multivariate data analysis (MVDA) and ML tools to build and validate advanced process models on complex bioprocessing data sets. Furthermore, delegates learn to apply advanced linear (e.g., PLS) and nonlinear (neural networks) techniques to analyze high-dimensional data sets such as those generated by process analytical technologies (PAT) devices (e.g., Raman spectroscopy). This module also incorporates aspects of big-data analytics to help delegates understand large manufacturing data sets and how to automate a model-building process. It emphasizes the importance of structured data storage to ensure that subsequent visualization and analysis can be automated. The module features hands-on activities using real bioprocess data sets leveraged from our industry–academia collaborations such as the FTHM Hub, enabling delegates to make informed data-driven decisions to complement their existing bioprocessing expertise and knowledge.
The Digital Skills Pipeline and Sustaining Growth of the Sector
In a previous article we argued that the training needs of the sector are two-fold: supporting the existing workforce with updates on new technologies and emerging therapeutic modalities while simultaneously establishing a pipeline of skilled individuals entering biopharmaceutical companies (5).
Developing our CPD modules to address the emerging skills needs of the bioprocessing industries serves as a guide for updating our undergraduate, master, and doctoral degree programs. In the near future, digital skills will be integral to the education of university graduates in the same way that genetic engineering techniques are embedded within bioscience programs. If new talent is to keep pace with the growth and diversification of the sector, it will be critical that educational programs prioritize digital skills training for larger student cohorts.
Figure 3 illustrates how UCL embeds digital skills into its degree programs. Graduates develop a systems-led approach to bioprocess design and research in which digital skills form integral parts of tools that students use to support problem identification, solution generation, and evaluation. To enter a sector manufacturing increasingly complex therapeutic products (e.g., viral vectors and vaccines), it is essential that students learn how to link the in silico and real worlds. UCL emphasizes integrating course material with practical modules and first-hand research experience.
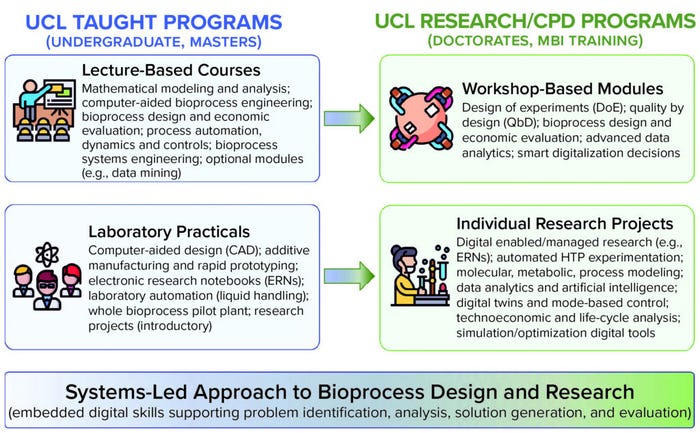
Figure 3: Digital skills are embedded within UCL biochemical engineering degree programs and progress from acquisition of digital knowledge and practical skills (undergraduate and masters programs) to their industrial application and further development (CPD and research programs). Programs include BEng/MEng Biochemical Engineering, MSc Biochemical Engineering, and MSc Manufacture and Commercialization of Stem Cell and Gene Therapies. Research degrees include PhD and industry collaborative EngD programs.
In our educational programs, students combine knowledge from core bioprocess engineering and mathematical modeling modules so they can synthesize and model entire processing flowsheets. Coursework culminates in a detailed bioprocess design project that includes economic and sustainability assessments for the manufacture of a specified therapeutic product. One recent example is when design groups focused on manufacturing COVID-19 therapeutics and vaccines. Complementary laboratory-based studies increasingly use electronic research notebooks (ERNs) and automated, high-throughput experimentation that urges students to think through digital data collection, storage, and analysis. We already use computer-aided design (CAD) and fabrication tools to exemplify principles of the engineering design cycle, rapid prototyping, and digital twins. Many training providers also are exploring parallel developments in virtual and augmented reality to enhance training experiences and outcomes.
In our research and CPD programs, we emphasize intensive short courses to provide specific digital skills that students/delegates can use in their research projects or workplaces. Some modules focus on acquisition of specific digital skills (e.g., experimental design or data mining) or use of specific software packages such as Python. Others highlight emerging digital technologies and how they affect critical business functions such as bioprocess validation, PAT, and quality-by-design (QbD) initiatives.
Ultimately, we want students to apply those skills in practice to test and refine digital predictions. Individual laboratory practicals increasingly expose students to digital technologies, automated experimentation, process control, and electronic data collection and storage. Students tackle increasingly open-ended and systems-based research projects as they progress from undergraduate to master and doctoral levels. The latter programs increasingly are enabled by digital technologies or focus on development of next-generation digital technologies (e.g., advanced data-mining algorithms and ML approaches).
Revolutionizing Bioprocess R&D
Digital technologies are set to revolutionize bioprocess research and development (R&D) and will underpin the introduction of Industry 4.0 concepts within the bioprocessing sector. A number of recent industry surveys have highlighted the interest of biopharmaceutical and bioprocessing companies in implementing digital technologies to improve efficiency, reduce costs, and increase flexibility. Results also highlight barriers to implementation, including an industry-wide lack of knowledge in digital technology and a difficulty in accessing suitably skilled individuals. UCL has evolved its training programs to meet the digital skills needs of the sector for both the short and long terms. Close interactions with industry professionals (e.g., through the FTHM Hub) ensure that our digital training provision is aligned to sector needs and contributes to the digital skills pipeline.
Acknowledgments
We gratefully acknowledge funding from the UK Engineering and Physical Sciences Research Council (EPSRC) for the FTHM Hub hosted at UCL with UK university partners. Financial and in-kind support from the consortium of industrial users and sector organizations also is acknowledged. Support for our CPD MBI modules at UCL Biochemical Engineering is gratefully acknowledged by Dr. Naveraj Gill (strategic alliance director) and Imogen Morehead (continuing professional development and “year in industry” program manager).
References
1 Branke J, Farid SS, Shah N. Industry 4.0: A Vision for Personalised Medicine Supply Chains? Cell Gene Ther. Ins. 2(2) 2016: 263–270; http://dx.doi.org/10.18609/cgti.2016.027.
2 Beckwith J, et al. Biopharma 4.0 — The Talent Continuum: Staying Afloat in Biopharma Talent Pool. Gen. Eng. News June 2022; https://www.genengnews.com/topics/bioprocessing/biopharma-4-0-the-talent-continuum-staying-afloat-in-biopharma-talent-pool.
3 Goran J, LaBerge L, Srinivasan R. Culture for a Digital Age. McKinsey Quarterly July 2017; https://www.mckinsey.com/capabilities/mckinsey-digital/our-insights/culture-for-a-digital-age.
4 Banner M, et al. A Decade In Review: Use of Data Analytics Within the Biopharmaceutical Sector. Curr. Opin. Chem. Eng. 34 2021: 100758; https://doi.org/10.1016/j.coche.2021.100758.
5 Gill NK, Lye GJ. Addressing Knowledge Gaps and Skills Development: Modular Training Keeps the Bioindustry at the Leading Edge. BioProcess Int. 15(9) 2017: insert; https://bioprocessintl.com/business/careers/modular-training-for-bioprocessing-industry-addresses-knowledge-gap-skills-development.
Corresponding author Suzanne S. Farid ([email protected]) is codirector of the Future Targeted Healthcare Manufacturing Hub, and a professor of bioprocess systems engineering. Duygu Dikicioglu is an associate professor of bioprocess digitalization. Stephen Goldrick is a lecturer of bioprocess digitalization. Gary J. Lye is a professor of biochemical engineering and head of the UCL department of biochemical engineering. All are at University College London, London WC1E 6BT, UK. MBI is a registered trademark of University College London.













