October 2015 Supplement
Just a few years ago, my requests for manuscripts detailing logistical considerations for cell-therapy manufacturing were met with puzzlement. The assumption seemed to be that such processes already existed for commercial biopharmaceuticals and would simply be adapted later. In quite a few cases, adopting a commercial mind-set did not appear to be a goal at all, with hands-on practitioners used to applying procedures in hospital settings.
For those of us who remember early discussions about commercialization and cost-containment for protein products, who watched the fledgling biopharmaceutical industry struggle through ups and downs in the 1980s–1990s, the question was how many existing procedures could be applied easily to cell therapies. Many would need major adaptation and rethinking. Since BPI’s first cell-therapy issue, we’ve watched as equipment and methods for cell growth and process scale-up benefited from existing technologies and began to diverge in ways specific to the needs of this emerging se...
Few other areas of medical research have been the source of as much promise (and hype) as cell therapies. Therapies that use engineered or repurposed versions of our own cells have inspired researchers and media alike. However, the pace at which effective therapies have made it out of laboratories and into clinical practice has not met the world’s high expectations. Although the biology has continued to press onward, the gap between R&D and commercialization remains substantial.
In 1957, the first human hematopoietic stem cell transplant occurred, transplanting bone marrow from one monozygotic twin to the other to treat leukemia (
1
). That eventually led to Joseph E. Murray and E. Donnall Thomas being awarded the 1990 Nobel Prize in physiology or medicine. By 1998, human embryonic stem cells were generated in the laboratory (
2
), sparking a flurry of research toward furthering our understanding of what has now become the field of regenerative medicine. As a result, cell therapy now occupies a prominent ...
The reprogramming of human somatic cells into induced pluripotent stem cells (iPSCs) offers tremendous potential for cell therapy, basic research, disease modeling, and drug development. Human iPSCs can be generated in culture, expanded, and then used to manufacture clinical-grade cells of almost any adult cell type. Given their great capacity for self-renewal, they are attractive as input materials for current good manufacturing practice (CGMP) operations.
For human iPSCs to fulfill their therapeutic potential, however, it is necessary to develop a CGMP- compliant iPSC manufacturing process and address challenges related to tissue sourcing, characterization, and testing of iPSC banks. Here, we summarize an approach to address the logistic and technical challenges involved in manufacturing clinical-grade cells. In addition to establishing a tissue- sourcing protocol, we developed a defined, feeder-free, and xeno-free reprogramming and cell culture system that supports efficient generation and expansion of...
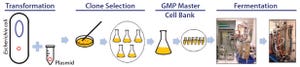
Figure 1: Development of production process for manufacturing plasmid DNA
DNA-based gene therapy products have been in clinical development since the 1990s. But over the past 24 months, the overall demand and therapeutic applications for plasmid DNA (pDNA) have rapidly grown and expanded. Currently, pDNA can be used directly as a therapeutic agent (e.g., in gene therapy or generation of vaccine antigens) and indirectly for a range of applications. Those include its use as a critical starting material for transient transfection to produce both viral-vector constructs (e.g., lentivirus or adenoassociated virus) and transient proteins in development (e.g., monoclonal antibody). Furthermore, pDNA is used as a master-template product to support production of new and emerging biopharmaceutical products and processes (e.g. RNA, in vitro protein production, and cell therapies).
Plasmids used as therapeutic agents tend to be relatively small in size (4.5– 6.0 kb and even down to 2.5 kb) (
1
), requiring relatively...
Planning for Commercial Scale of Cell Therapy and Regenerative Medicine Products, Part 2: Clinical Efficacy, Reimbursement, and Needle-to-Needle LogisticsPlanning for Commercial Scale of Cell Therapy and Regenerative Medicine Products, Part 2: Clinical Efficacy, Reimbursement, and Needle-to-Needle Logistics
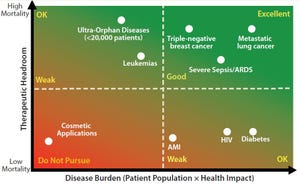
Figure 1: Assessing your therapies’ reimbursement potential
Cell therapy is an emerging pillar in healthcare with the potential to provide curative solutions to a wide range of indications. The biological complexities through which cell technologies exert their clinical impact (especially those used in immunotherapies for cancer) provide opportunities for novel modes of immune regulation, cell targeting, and payload delivery. Cells also can serve as vehicles for genetic content, which the gene therapy industry is now investigating.
Since early 2004, Invetech has worked with organizations dedicated to cell therapy and regenerative medicine to help them develop and implement commercial-scale manufacturing for a broad range of therapies. In part 1 of this two-part article (
1
), we introduced the elements for building a solid foundation for game-changing cell-therapy product development. We call them the “five pillars of success”:
Part 1 focused on the first two pillars of success (manufacturability and cost...
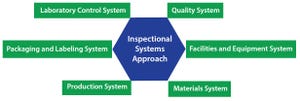
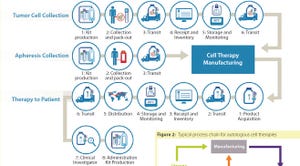
Commercializing cell therapies can be much more challenging than commercializing traditional pharmaceuticals and biologics. Cell-based drug products are significantly more complex than protein or small-molecule drug products. Their mechanisms of action and product attributes are also more complex. Cell therapy product attributes rely heavily on the associated manufacturing processes. Process changes can influence products in ways that may not be discernible until their effects on efficacy effects become evident. Product characterization is critical, but cell therapy products are living organisms with many characteristics. For pharmaceuticals and biologics, planning and process changes for commercial manufacturing often can wait until clinical results are available. But for cell therapies, changes during planning and processing carry a much higher risk. Companies may be unable to determine the effects of such changes on product efficacy without needing more clinical data.
As a result, planning a cell thera...
Photo 1: Shipping containers
Among the basics of building a successful logistics strategy for the management of cell-based material, some better-known and important factors to consider include selecting the right dry-shipping unit, qualifying that container for a particular payload and shipping configuration, choosing an appropriate data logger, creating a chain of custody, evaluating a transit carrier, and anticipating potential problems inherent in shipping at cryogenic temperatures (
1
). Here, I’d like to go beyond those basics to address some lesser-known considerations. These factors may be hidden in the background, but they can play a major role in the success or failure of a clinical trial as well as the long-term efficacy of a cell-based commercial product. They include standardization, package and shipping qualification, equipment validation, process qualification, and documenting the chain of custody.
Several unique logistical challenges arise for autologous cell therapies — those that use a pa...