Voices of Biotech
Podcast: MilliporeSigma says education vital to creating unbreakable chain for sustainability
MilliporeSigma discusses the importance of people, education, and the benefits of embracing discomfort to bolster sustainability efforts.
Sponsored by Sartorius
Biopharmaceutical developers and manufacturers are part of a global, dynamic, and highly competitive market. They face constant pressure to produce high-quality products within relatively short time frames and at reduced costs. Process-intensification strategies and single-use (SU) solutions are popular approaches to maximizing productivity and promoting fast, efficient, and lean processing — the pillars of next-generation facilities.
Filtration is an integral part of all bioprocesses and is applied to many up- and downstream steps, including harvest, clarification, and concentration/diafiltration. Tangential-flow filtration (TFF) is one method that represents a valuable opportunity for improvement. The increased speed and titers generated by intensified processing put additional strain on TFF operations. TFF has diverse applications, including isolating a product of interest while eliminating contaminants, exchanging buffer solutions in preparation for subsequent chromatography steps, and reducing volume.
Here, we consider how adopting SU TFF can support faster and more productive bioprocessing. We describe how Sartorius hollow-fiber TFF modules offer particular advantages and contribute to meaningful reductions in processing time.
The Value of TFF
Typical filtration methods (sometimes referred to as normal-flow filtration or direct-flow filtration) rely on passing a liquid through a membrane to isolate components of interest from a complex mixture. Fluid flows perpendicularly to the membrane, and the filter collects impurities as a sample volume passes through it. However, that approach can create clogging and shearing, which is of particular concern with viscous solutions and shear-sensitive molecules.
TFF solves some of those issues. In TFF (also known as cross-flow filtration) a liquid passes parallel to the surface of a filtration unit. Small molecules pass through the filter as part of the permeate, whereas larger molecules recirculate back to a feed reservoir. The direction of flow limits membrane fouling and enables processing of larger sample volumes.
Sartorius offers two TFF technologies: flat-sheet cassettes and hollow fibers. Herein, we focus on hollow-fiber modules: cylindrical units formed by bundles of individual fibers composed of semipermeable membranes. Filtration occurs when a sample passes through the fiber membrane (permeate). The solution passing along the membrane is circulated back to the feed vessel (retentate). In hollow fibers the open-channel flow paths contribute to a relatively gentle separation, causing less shear stress to a product of interest.
Single-Use TFF: A Solution to Industry Problems
Biopharmaceutical developers require robust tools that support rapid separation and high recoveries to improve filtration efficiency and maximize yields – even for emerging, sensitive modalities (e.g., large enveloped viruses, extracellular vesicles, and viral vectors). Ready-to-use, reproducible solutions are sought to improve batch-to-batch consistency and enable clearance of regulatory hurdles. Next-generation SU TFF solutions are needed to address many challenges associated with modern bioprocessing and keep up with dynamic industry demands.
Poor Filtration Efficiency in Cell Harvest and Clarification: Conventional SU clarification tools cannot fully clarify microbial fermentation broths and high-density mammalian cell cultures, leading to poor efficiency and low recovery. By contrast, hollow-fiber TFF modules are more tolerant of fluids carrying high levels of solids, making such modules ideal for use in harvest and clarification steps for high-biomass samples created by intensified upstream processing. That improved efficiency allows a greater sample volume to be processed per membrane area, increasing filter capacity and process speed. Ready-to-use Sartorius hollow-fiber TFF modules incorporate a robust, macro-void free membrane and come in sizes from 52 cm2 to 15.42 m2, enabling scalable processing from 20 mL up to thousands of liters.
Low Recovery of Emerging Modalities in Downstream Processing: New modalities often are more susceptible to damage by shear forces than are traditional biotherapeutics such as recombinant proteins. Hollow-fiber TFF offers a spacer-free, open-channel flow path to create a gentle environment for fragile biologics, improving purity and yield without compromising efficiency. Sartorius offers hollow-fiber modules with high molecular-weight cut-off (MWCO) options up to 750 kD, as well as a range of hollow-fiber microfiltration membranes from 0.1 µm to 0.65 µm.
Time-Consuming Equipment Preparation and Validation: Multiuse filtration setups require cleaning and validation to prevent cross-contamination. Such time-consuming, costly, and labor-intensive procedures require increased operator involvement, which can introduce variability into a process. Multiuse equipment also requires lifetime testing for membrane reuse to determine its performance and durability across multiple process runs (1). Product-specific lifetime studies must be carried out to ensure that tested conditions reflect the activities involved in each bioprocess.
Implementing SU equipment eliminates the need for certain cleaning validation steps. The Sartorius SU hollow-fiber filters come in fully self-contained and gamma-irradiated assemblies and are designed to fit individual process needs. In addition, they are free from humectants (e.g., glycerol) and do not need to be flushed before use; a small-volume buffer equilibration is sufficient. Thus, the modules truly are ready to use, making their implementation faster, safer, and more convenient. That in turn frees up valuable operator time and shortens the total process duration for greater use of production suites.
Meeting Regulatory Compliance: Scalable, robust, consistent, and reproducible operations are essential for obtaining regulatory approval. SU setups do not require cleaning validation, improving their overall reliability. That eliminates a source of variability in biopharmaceutical processes and simplifies regulatory applications.
Adaptability: Finding the Right Solution
Each process is unique; filtration solutions should reflect that reality and adapt to fit the requirements of each facility and modality. SU tools often offer such flexibility. Sartorius SU hollow-fiber products enable bioprocess scientists to design comprehensive customized assemblies that provide all components needed for fully integrated membrane separation solutions for their systems. CrossflowExpert calculators support decision-making and help users find the correct TFF modules and operating conditions for their applications.
SU TFF: Tangible Time Savings
TFF is used throughout bioprocesses, including in clarification steps, before and/or between chromatography steps, and during concentration and diafiltration steps to perform final product filtration. Thus, the importance of maximizing the performance and speed of TFF unit operations cannot be overstated.
Sartorius SU hollow-fiber TFF modules incorporate a robust membrane that is produced internally, removing a source of potential variance in product quality. Compared with reusable setups, SU TFF can save up to 50% of facility time by eliminating rinsing, integrity testing, normalized water permeability (NWP) testing, cleaning, consumable removal, and storage operations (Figure 1).
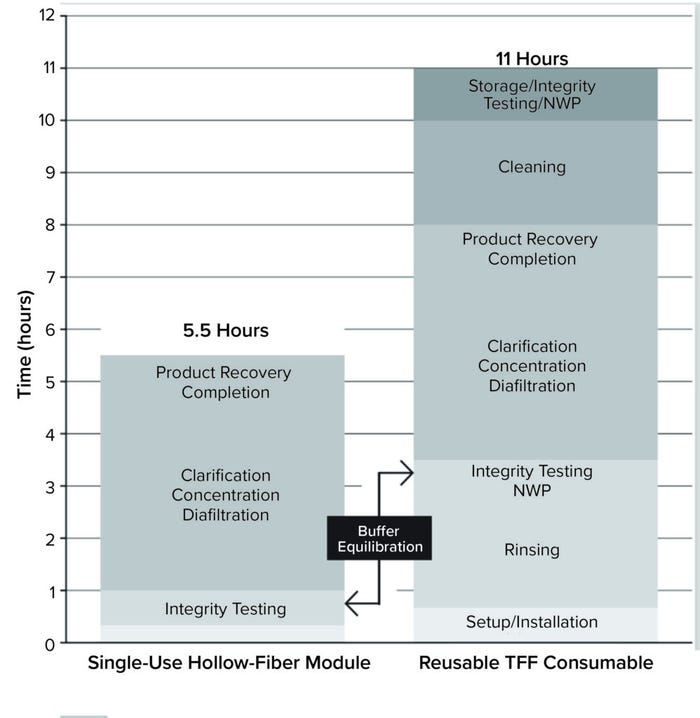
Figure 1: Filtration with Sartorius single-use hollow-fiber modules is significantly faster than with reusable tangential-flow filtration (TFF) consumables (NWP = normalized water permeability).
In addition to their suitability in intensified and continuous operations, the related time savings make Sartorius SU hollow-fiber TFF modules ideal for use within eight-hour shifts, which is an essential consideration for day-to-day facility operations. Ultimately, the increased capacity offered by TFF and additional benefits created by incorporating sustainable SU solutions will occupy an increasingly important place in bioprocessing facilities of the future. Similar performance, time savings, and quality advantages can be achieved when implementing Sartorius’s broader SU portfolio of downstream processing solutions.
Discover more at https://www.sartorius.com/hollow-fiber-tff, and configure your solution using CrossflowExpert calculators at https://watersep.net/crossflowexpert.
Reference
1 Rathore AS, et al. Lifetime Studies for Membrane Reuse: Principles and Case Studies. BioPharm Int. 20(9) 2007: 48–54.
Franziska Froboese is a product manager, [email protected]; Katy McLaughlin is a scientific content writer, [email protected]; Bengt Persson is global product specialist for hollow-fiber TFF, bengt [email protected]; and Marcel Fueger is product manager, support, [email protected], all at Sartorius in Göttingen, Germany.
You May Also Like