- Sponsored Content
- Laboratory Equipment
- Product Characterization
Speeding Characterization of Biologics: Replace Traditional Assay Technologies with Label-Free Quantification and Kinetics
December 14, 2018
Sponsored by Forte Bio
 FortéBio’s Octet instruments are an ideal replacement for ELISA, HPLC, and SPR techniques in quantification of antibodies and recombinant proteins and in testing product potency for lot release. Bio-Layer Interferometry (BLI) technology monitors biomolecular interactions in real time to determine affinity, kinetics, and concentration. The plate-based, microfluidics-free format offers users several distinct advantages over other technologies. BLI-based systems can achieve higher throughput, with the flexibility to measure two to 96 samples simultaneously. Lower maintenance requirements and increased ease-of-use further shorten the time it take to get results. Increased system flexibility — including an ability to analyze unpurified samples and tolerate a broad range of conditions — is another time-saving hallmark. With additional tools to enable integration into regulated environments, the Octet systems can be instituted across biologics development.
FortéBio’s Octet instruments are an ideal replacement for ELISA, HPLC, and SPR techniques in quantification of antibodies and recombinant proteins and in testing product potency for lot release. Bio-Layer Interferometry (BLI) technology monitors biomolecular interactions in real time to determine affinity, kinetics, and concentration. The plate-based, microfluidics-free format offers users several distinct advantages over other technologies. BLI-based systems can achieve higher throughput, with the flexibility to measure two to 96 samples simultaneously. Lower maintenance requirements and increased ease-of-use further shorten the time it take to get results. Increased system flexibility — including an ability to analyze unpurified samples and tolerate a broad range of conditions — is another time-saving hallmark. With additional tools to enable integration into regulated environments, the Octet systems can be instituted across biologics development.
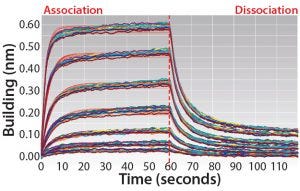
Figure 1: Overlay of several replicates of FcRn–IgG interactions on the Octet platform with FAB2G biosensors. Data-trace fitting used a 1:1 model with global fitting and a 5-second dissociation step (fit lines in red).
Applications
Concentration Determination: For measuring antibody and protein concentrations, the Octet platform provides a convenient and reliable Dip and Read approach. Rapid quantitation of 96 samples can be completed in ≥5 min on an Octet HTX system (80 samples in <30 min with an Octet RED96e system). Concentrations can be measured directly in cell culture supernatant or lysate, eliminating the need for purification.
High-Precision Ligand Binding and Potency Assays: Ligand-binding kinetics assays are used increasingly in batch lot release, especially for potency assays for assessing product stability and function. In binding-kinetics studies, interactions often
are assessed through measuring either the affinity of an analyte (e.g., drug product) to a receptor or ligand immobilized on the biosensor surface or by monitoring the analyte‘s binding response signals as a function of concentration and comparing results with a control reference for relative potency assessment. Octet systems high-throughput capability and plate-based format enables users to create design of experiments (DoE) that allow for rapid assay optimization, hence remarkably reducing the time to results. In addition, these systems’ ease of use allows for easy transfer of methods into quality control (QC).
Stability and Forced-Degradation Studies: Octet platforms also can be used for developing stability-indicating methods. They are suitable for measuring and distinguishing between fully functional drug products and those for which binding activities have been affected by degradation.
Analytical Instruments in the GXP Environment
Analytical methods that provide reliable and accurate data are necessary to ensure that quality standards are met during production and release of drug products. To ensure data accuracy, it is critical that the performance of analytical instruments, computer systems, and methods of data acquisition meet required specifications. FortéBio offers software, qualification kits, and services to assist in installing and maintaining Octet instruments for regulated environments:
Octet CFR software for secure, traceable electronic recordkeeping to enable compliant data acquisition and data analysis
IQ/OQ/PQ packages to ensure that Octet systems are qualified and operate as intended and to meet specifications
Customer-run software validation support
Biosensor validation support services
Technical support assistance.
Octet high-throughput characterization systems can accelerate development programs for biologics and increase the data available for decision making throughout the entire workflow. With complete qualification, validation, and support solutions for regulated laboratories, the Octet platform can replace traditionally time-intensive techniques.
David O. Apiyo, PhD, is marketing applications manager at FortéBio – Biologics by Molecular Devices, ForteBio LLC, 47661 Fremont Boulevard, Fremont, CA 94538; 1-650-322-1360; [email protected].
You May Also Like





