Martin Smith, PhD
CTO, Pall Corporation
Thank you for taking the time to view our supplement, which has been driven by our desire to share actionable knowledge in a convenient format as well as to act as a guidepost for the evolution of continuous processing. As you read, we encourage you to ask questions, share your thoughts, or send general input/requests to our team at www.pall.com/ continuous-questions.
We hope that this supplement encourages a deeper discussion about continuous bioprocessing technology and the dramatic impact it can have — and already is having — on the biopharmaceutical industry. Beginning with a look back at the history of continuous processing in other industries and moving forward to the potential it has in the bioprocessing industry, we move into how Pall is working to continuously improve bioprocesses. Our special issue closes with projections for the future.
Thank you to everyone who participated in the creation of this supplement, whether directly or indirectly. The editorial...
Continuous and semicontinuous manufacturing systems have been used for many years in numerous sectors — including the automotive, food and beverage, oil refining, chemicals, pulp and paper, electronics, metal smelting, steel making, and waste-water treatment industries. Most of these industries are capital intensive and switched to flow manufacturing to increase productivity and flexibility; reduce cycle times, inventory, waste, and costs; and achieve enhanced product quality. Despite the capital intensive nature of drug manufacturing, the biopharmaceutical industry has lagged behind these other sectors in implementing continuous, or even semicontinuous processing.
Drivers for Continuous Processing In Nonpharma Sectors
The reasons for the switch from batch to continuous (or semicontinuous or line-flow) manufacturing in various industries differ, but there are some commonalities. The production of pig iron in blast furnaces is one of the oldest examples of a continuous process. The paper industry moved to...
Numerous industries have demonstrated that continuous manufacturing provides significant benefits and advantages, ranging from reduced capital and operating expenses to greater efficiency and product quality and consistency. The conventional approach to biopharmaceutical manufacturing involves the batch performance of a series of unit operations separated by hold steps requiring additional tanks and/or biocontainers. Such an approach fails to maximize facility use, requires large buffer volumes, and results in overall inefficiencies.
An integrated, continuous approach to bioprocessing connects each unit operation, minimizing the need for intermediate hold steps. As a result, the overall process is more efficient, has a smaller physical and environmental footprint (favorable process mass intensity, PMI, values and environmental factors), requires less initial capital investment, and has lower overall operating costs. The longer operating periods (weeks or months compared with hours to a day or two) also me...
Martin Smith, PhD, Chief Technology Officer, Pall Corporation
Martin Smith, PhD, has been with Pall for about nine years and assumed the role of chief technology officer at Pall about 18 months ago. He spoke with Cynthia Challener, PhD, about Pall’s biopharmaceutical business unit and how the company is positioning its technology suite for a continuous process paradigm.
Smith:
There is no doubt in our minds that we see movement toward continuous bioprocessing. When you look across an array of different industries, the move to continuous or parallel processing is paramount. A simple example of a unit operation is collecting water from a well rather than through the continuous process of turning on a faucet. When pulling water from a well, you may have variations in the way different operators pull that water up. They may spill some. They may need to travel some distance. Similarly, in the biopharmaceutical industry today we have clusters of individual unit operations that are just sort of bolted together ...
Regardless of the industry and product being manufactured, continuous processing has demonstrated numerous benefits. In addition to smaller manufacturing footprints, reduced material consumption and waste generation, increased efficiencies, and lower capital and operating costs continuous manufacturing typically leads to more consistent processes and product quality. In the pharmaceutical industry, the latter two attributes align perfectly with FDA’s Quality by Design (QbD) and process analytical technology (PAT) initiatives. The challenge is determining how to apply these concepts in practice.
Applying the Idea of a Batch/Lot to Continuous Processes
Because the pharmaceutical industry has traditionally used batch-based manufacturing processes, regulatory requirements pertain to batches or lots, as outlined in the various regulations and guidance documents from agencies around the world such as those in ICH Q7, 21 CFR 210, Health Canada’s GMP guidelines (GUI-0001), and EMA MEDDEV 2.5/6 Rev. 1.
In general...
Michael Egholm, PhD, Vice President and General Manager of Biopharmaceuticals at Pall Life Sciences
During the March 2016 BPI West conference in Oakland, CA, BPI publisher Brian Caine and I had an opportunity to meet with Michael Egholm, PhD, Vice President and General manager of Biopharmaceuticals at Pall Life Sciences. We are pleased to be able to share his thoughts about Pall’s support of continuous processing — and the company’s current offerings.
Montgomery:
Not everyone seems to be defining continuous processing in the same way. What is your general concept of it?
Egholm:
At Pall Life Sciences, we see continuous processing (and the pun is intended here) as a continuous evolution of the life sciences industry.
Over the past decade, the transition to single-use technologies has taken a great deal of the spotlight, reaching an inflection point where we now see very rapid growth and adoption. Continuous processing is the next logical extension of that evolution because it aims to link technologies to ...

 +1
+1Unlike chemically synthesized drugs, whose structure is known and reproducible, biological drugs are derived from living cells and are sensitive, complex mixtures requiring cutting-edge biological technologies for their production. The growing importance of biosimilars in recent years is reflected in a corresponding rise in market value. The value of the global biologic therapeutic drug market reached approximately US$230 billion in 2014 and, according to BCC Research, will increase to nearly $390 billion by the end of 2019. This corresponds to a compound annual growth rate (CAGR) of just over 10%. BCC Research also estimates that biosimilars accounted for nearly $2.0 billion of the biologic therapeutic drug market in 2014 and that this segment is growing at a CAGR of 15%. Key drivers for the growth of biologic therapeutic drugs include the aging of the global population and the worldwide increase in chronic diseases; biologic drugs are very effective for treating arthritis, diabetes and heart diseases. R...
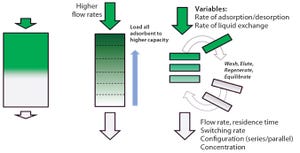
Figure 1:
Principles of simulated moving bed technology
Intensified and integrated bioprocess technologies are creating a paradigm shift toward more efficient, higher flexibility facilities for biopharmaceutical manufacturing. Continuous technologies that are designed as single-use systems help to greatly facilitate process intensification, delivering further efficiencies with reduced set-up times and elimination of the need for cleaning and cleaning validation.
Chromatography is often considered to be a challenging bioprocess step, which has caused great interest in a simplified, safer solution. Continuous multicolumn chromatography using a single-use flow path is an economically viable, disposable chromatographic solution for even the most demanding and costly chromatographic steps in a manufacturing process.
The Cadence BioSMB PD platform from Pall Life Sciences is the first disposable flow path, continuous multicolumn chromatography solution that is scalable from a process development (PD) laboratory...

 +1
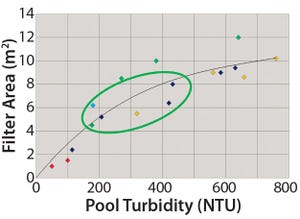
+1Figure 1:
Depth-filter sizing following AWS
processing (1,000-L batch)
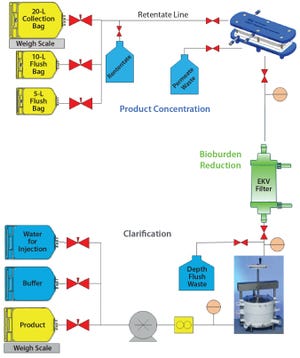
Current bioprocesses combine fed-batch cell culture with batch-wise downstream processing steps. To achieve integrated upstream and downstream continuous manufacturing, the industry has been in need of a continuous cell separation and clarification solution for bioprocess fluids from bioreactors. The Cadence Acoustic Separator from Pall Life Sciences provides this solution, with continuous first-stage clarification without the need for filter media in a scalable, single-use format with no negative impact on product attributes. The Cadence Acoustic Separator delivers cost and time savings through reductions in buffer volumes, filter preparation time, and depth-filter requirements with stable performance and higher yields over longer processing times.
Limitations of Existing Technology
As the biopharmaceutical industry slowly but surely moves toward adoption of integrated continuous manufacturing, suppliers such as Pall are driven to d...
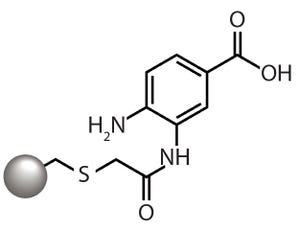
Figure 1: The aminobenzoic acid ligand of
the CMM HyperCel mixed-mode cation
exchanger provides cation-exchange
functionality through the carboxyl group and
hydrophobicity-based binding through the
aromatic ring.
The biopharmaceutical industry is under a great deal of pressure to modernize manufacturing to meet the challenges of production at vastly different scales for niche drugs as well as for expected massive blockbusters, biosimilars, and regional manufacturing. To address these challenges, the biopharmaceutical industry is embracing process intensification through single-use and continuous processing technologies. Implementing these technologies offers increased productivity and manufacturing flexibility and reduces the footprint, capital outlay, and operating costs.
Pall Life Sciences has developed several technologies designed to enable single-use and continuous processing for a broad range of downstream unit operations. The Pall systems are designed to be interconnected to offer not just increase...

 +1
+1Pall’s Center of Excellence, Westborough MA
Mario Philips is Vice President and General Manager of Single-Use Technologies at Pall Life Sciences. In February, he spoke with BPI publisher Brian Caine and editor in chief Anne Montgomery about Pall’s commitment to enabling continuous processing and its development of single-use technologies. In that discussion, he addressed some major process bottlenecks and Pall’s solution to them, including centrifuge replacements by continuous acoustic wave separation, continuous chromatography with multicolumn chromatography technology platform, and a simplified version of tangential-flow filtration.
Read the full text of this article in the PDF (Login required).

 +1
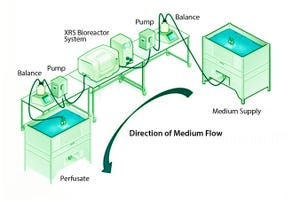
+1Although implementation of continuous manufacturing for biopharmaceuticals is in the early stages, continuous cell culture technology has been around for close to thirty years. Perfusion was initially developed in the late 1980s as a means for increasing protein titers (
1
). However, high costs driven by media consumption limited widespread commercial adoption. In the same time frame, advances in cell line engineering, media composition, and bioreactor design led to 10-fold increases in titers for batch and fed-batch modes, eliminating the first driver for adoption of perfusion technology. As a result, over the next 20 years or so, perfusion processes were largely used only for production of toxic or unstable proteins that degrade if exposed to culture conditions for typical batch/fed-batch residence times. Recently, however, growing recognition in the industry that there is a need for alternative manufacturing strategies that can boost efficiency and productivity while reducing costs has led to renewed ...
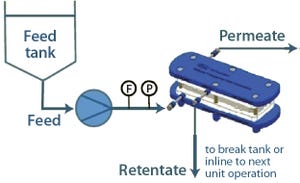
Continuous processing has the potential to provide significant cost and time savings for biopharmaceutical manufacturing, but that potential can be realized only if appropriate automation solutions are available for continuous flow between disparate upstream and downstream operations. Pall Life Sciences’ Allegro MVP system, a fully automated bioprocessing system designed for use in upstream and downstream single-use processing, enables flexible automation and thus facilitates continuous biopharmaceutical manufacturing. This article presents the results obtained using the Allegro MVP system in combination with the Cadence Inline Concentrator (ILC) to demonstrate the system’s depth of flexibility.
About the Allegro MVP System
Automation combined with single-use technologies provides additional benefits during manufacturing, including consistency in product quality and reduced labor costs and operator errors. To realize these benefits for a multitude of unit operations in bioprocesses, Pall designed the Alle...

 +1
+1






.jpg?width=700&auto=webp&quality=80&disable=upscale)

