April 2016 Supplement
In many ways, patient-specific cell therapies (PSCTs) are still the “new kid on the block” in medicine. Researchers, therapeutic developers, manufacturers, regulators, and payers are still exploring and developing an understanding of the powerful benefits and unique challenges associated with this growing industry. As we all become more familiar with PSCTs, an evolution will need to occur — as it has for automobiles, computers, and every technological advance in human history — for these therapies to become widely adopted, cost-efficient, market-scalable, and sustainable over the long-term. From where we are now as an industry, what is needed from a manufacturing perspective for us to achieve a manifest future for patient-specific cell therapy?
Table 1: Differences between traditional biologics and PSCTs; product attributes in green, manufacturing design drivers in blue
Not Your Grandfather’s Medicine
From their compositional complexity to their mechanisms of action to their manufacture, PSCTs are very di...
Figure 1: Organizations developing AAV vector candidates
Recent advances in molecular biology are expediting genomic sequencing to underpin precision medicine. Such progress is positioning gene and gene-modified cell therapy on the cusp of an extraordinary revolution in patient care for presently unmet medical needs — and a new therapeutic class that could rival monoclonal antibodies (MAbs) in importance. However, despite substantial strides made in clinical trials, the bioprocessing community is struggling to fulfill growing demands for biomanufacturing capacity to make gene and gene-modified cell therapies — including current good manufacturing practice (CGMP) viral vectors.
Here we review the basic science of gene therapy bioproduction and evaluate critical emerging bioprocess opportunities and challenges. An interrelationship exists between viral-vector upstream and downstream bioprocessing strategies and those for other therapeutic platforms such as chimeric antigen receptor (CAR) T cells and induced...
© INVETECH 2016 (www.invetech.us)
The concept of automation conjures up images of robots on assembly lines or perhaps automobiles replacing horse-drawn carriages. In both examples, automation provides an ability to work tirelessly, with reproducible high-quality outputs at increased speed. For cell therapy, automation can be used to increase the scale of cell culture operations (e.g., bioreactors replacing flasks) and allow the use of closed systems that can protect cell products from contamination with adventitious agents from the environment or operators themselves. Closed systems also can protect a facility from product-related contaminants or viral reagents and reduce the risk of cross contamination between products made in a shared facility.
Automation can improve analytical testing that supports manufacturing and can facilitate quality control by reducing operator variability and subjective judgment of results (e.g., based on visual interpretation). Automation allows for continuous monitoring of pro...
Continued clinical efficacy demonstrations of cell-based immunotherapies (iTx) such as chimeric antigen receptor T cell (CAR-T) therapies has made the prospect increasingly likely of an immunotherapy product achieving conditional market authorization in the short term. For example, Novartis and the University of Pennsylvania’s lead candidate (CTL019) for treating a range of hematological malignancies received breakthrough status from the US Food and Drug Administration (FDA) in 2014, permitting access to an expedited drug development pathway for high unmet medical needs (
1
). Then in March 2015, the European Medicines Agency’s (EMA’s) Pediatric Committee agreed on a pediatric investigation plan for tisagenlecleucel-T (
2
).
Figure 1: Methods for producing genetically modified T-cells for immunotherapy include (A) T-cell receptor obtained from a patient with the disease; (B) T-cell receptor obtained by immunizing transgenic mice expressing human major histocompatibility complexes (MHCs) with target cancer...
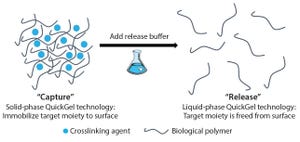
Releasing and separating cells from surfaces and capture molecules are critical steps in cell therapy development. Research into such therapies as chimeric antigen receptor T cells (CAR-T) cancer therapies and stem-cell regenerative medicines demand the isolation and purification of viable and functional target cells. A number of cell-separation strategies can be used to produce such cells, but they are not able to deliver the required efficiency or scalability and can also cause damage to cells or affect their phenotype.
Figure 1: QuickGel hydrogel platform
Since its foundation in 2013, Quad Technologies has focused on its platform QuickGel technology, a dissolvable “cell friendly” hydrogel (Figure 1). In late 2015, the company also launched its MagCloudz streptavidin cell-separation kit and partnered with the University of Massachusetts Medical School in research toward the enrichment and purification of CD3+ T cells from human umbilical cord blood. The collaboration is part of the company’s overall pla...
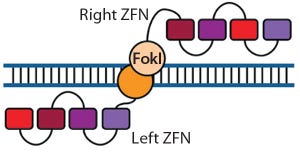
Historically, fundamental science and process engineering were separated by distinct vernaculars and a decade or more in the translation pathway of candidate therapeutics from laboratory to bedside (
1
). This crude metric holds true for the origins of the modern pharmaceutical industry, namely fine chemicals that supported the high-margin small molecules that constitute the majority of the pharmacopoeia even today. But as illustrated by deeply interwoven careers, companies, and technologies — including those related to monoclonal antibodies (MAbs) — that classic gap between science and bioprocessing is rapidly closing (
2
). Arguably, therapeutic gene editing technologies could represent the greatest stimuli yet to span this chasm conclusively and build networks for future emerging technologies.
Therefore, the
raison d’etre
of this article is simple: to urge the global bioprocessing community to recognize therapeutic gene editing as not simply a scientific curiosity to adorn the ephemeral pages of
Nat...
Xuri cell culture media are formulated for the growth and expansion of human mesenchymal stem cells after their isolation from a number of sources.
With support from the federal government of Canada, GE Healthcare and the Centre for Commercialization of Regenerative Medicine (CCRM) are pushing into new frontiers to advance the progress of cell therapy and regenerative medicine. When I first met Michael May, president and chief executive officer (CEO) of CCRM, both our organizations had been exploring opportunities in parallel to drive the cell therapy industry forward. CCRM’s mission is to create and sustain a global nexus for company creation, technology and cell therapy development, and clinical trials in regenerative medicine. The goal of GE’s cell therapy business was to introduce solutions and tools to the cell therapy industry and thus enable scalable, commercialized manufacturing of such therapies. Combining our common interests and complementary expertise seemed opportune.
With a $40 million (Cana...