Chromatographic Purification in Downstream ProcessingChromatographic Purification in Downstream Processing

In many biopharmaceutical companies, chromatographic purification in downstream processing is a key focus for optimization studies. The objective is to streamline the process, which may be achieved by the elimination of intermediate unit operations. The main question, therefore, is how to optimize the adjustment of individual chromatographic process steps.
Currently there is a broad range of next-generation sorbents and membranes available for process chromatography. These new chromatography media are characterized by significantly improved performance compared to their classical predecessors: e.g., higher dynamic binding capacities, higher operational flow rates, and specific and distinctive retention mechanisms. Nevertheless, traditional media (based on agarose or polymers) continue to be routinely used for new method development due to their proven suitability for protein purification in FDA- or EMA-approved applications. In these cases, the potential of modern ion exchangers and novel mixed-mode or multimode sorbents still remains poorly exploited. As a result, significant additional costs in production may be incurred. Practical experience shows that the optimization of individual process steps (capture, purification, and polishing) already offers important cost reductions. But it is mainly through a combination of all sorbent- and membrane-based unit operations deployed in downstream processing that the door is opened for effective process cost savings.
Ion-Exchange Chromatography
Anion and cation exchangers have been among the most important tools in protein purification. Ion exchange is well known by regulatory authorities and is a user-friendly technique. Recently, new sorbent manufacturing processes have significantly improved their capacity and performance. An example is Pall’s Q HyperCel™ and S HyperCel ion-exchange sorbents, which were designed with differentiated selectivity for the purification of proteins at laboratory, pilot, and full manufacturing scales.
Q HyperCel and S HyperCel sorbents exhibit high dynamic binding capacities at high flow rates (and low residence times such as 1 or 2 minutes, as in Figure 1), and enable increased throughput.
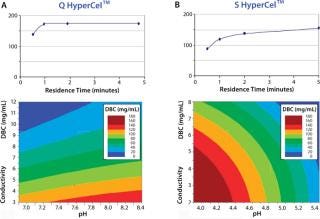
Figure 1: ()
Mixed-Mode Chromatography
New perspectives for biopurification are also provided by sorbents that interact with the target protein by a multiple retention mechanism. Mixed-mode MEP, HEA, and PPA HyperCel sorbents’ binding mechanism is primarily a combination of hydrophobic and/or pseudoaffinity interactions with the target. Typically this is achieved without additional modification of the feedstock. MEP HyperCel is uncharged under typical loading conditions (pH ~7), whereas the majority of proteins carry a net negative charge. At acidic pH, both MEP ligand and bound proteins take on a net positive charge. Elution is induced by electrostatic repulsion, by decreasing the pH. HEA and PPA HyperCel sorbents carry different ligands (aromatic and aliphatic) and operate under similar conditions, but offer a different selectivity. Recently, process developers have shown that the addition of 0.1–0.5 M arginine to MeP HyperCel sorbent elution buffers allows for protein elution at pH values around neutrality (arakawa et al., 2009).
Two-Step Antibody Purification
Monoclonal antibody purification traditionally implies a protein a capture step followed by two orthogonal steps. According to the antibody, the sequence and type of steps after protein A may vary. But generally, intermediate unit operations are required such as diafiltration for buffer exchange or addition of salt for hydrophobic interaction.
An alternative two-step purification process is based on the use of MEP HyperCel sorbent for the primary capture and S HyperCel sorbent for the second chromatographic step (Ferreira et al., 2007). These two sorbents are complementary to each other and assure both yield and purity of the target. Requirements for intermediate unit operations are eliminated for better process economics. Non-protein A purification schemes, including MEP HyperCel at intermediate step following Mab capture by cation exchange, have also been recently reported (Conley et al., 2011).
Membrane Chromatography
Typically, the capture and intermediate chromatographic steps are in positive mode (the target molecule is bound to the sorbent). However, to ensure the remaining level of contaminants (DNA, HCP, endotoxin, and virus) meet regulatory approval, the final chromatographic step tends to be in negative mode. In this mode, contaminants are bound, and the target is in the flow through. Chromatography membrane adsorbers are ideally suited for negative mode or polishing applications. A significant advantage of membranes over classical sorbents is their larger pore size, which allows enhanced accessibility for macromolecules (e.g., DNA and viruses) and allows for faster processing using much higher flow rates than packed-bed resins. Membrane adsorbers are available in disposable capsule formats that eliminate the need for column packing and cleaning. The family of Mustang® XT membrane adsorbers with scalable formats meets all requirements from laboratory-scale to process-scale applications.
Process Hardware
A reproducible and robust manufacturing process under GMP conditions requires high-performance columns (Figure 2) and control skids to ensure the performance of the validated process. Flexible and robust Resolute® columns and packing stations in combination with PK systems for process-scale chromatography and PKP systems for pilot-scale chromatography offer the optimal hardware platform for all types of sorbent or membrane-based operations.

Figure 2: ()
About the Author
Author Details
Dr. Dirk Sievers is marketing
manager at Pall GmbH Life Sciences, Dreieich BioPharmaceuticals Central Europe; 49-6103-307-582, fax 49-6103-307-295; [email protected]. Dr. Sylvio Bengio is marketing chromatography and scientific communications for Pall Life Sciences, Cergy France; 33-1-3420-7823; [email protected]; www.pall.com/biopharm
You May Also Like






