- Sponsored Content
Reimagining Capacity for Today’s Purification of Monoclonal AntibodiesReimagining Capacity for Today’s Purification of Monoclonal Antibodies
January 26, 2018
Sponsored by GE HealthCare Technologies
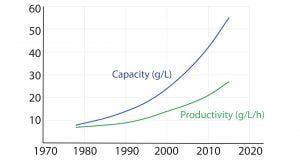
Figure 1: Protein A chromatography resin capacity and productivity (adapted from Bolton G, et al. The Role of More Than 40 Years of Improvement in Protein A Chromatography
in the Growth of the Therapeutic Antibody Industry. Biotechnol. Prog. 32, 2016: 1193–1202)
The monoclonal antibody (MAb) market has grown over the past decade to be about half of the biomanufacturing market today. This growth should continue, driven by a strong pipeline of MAbs that are currently in phase 2 and 3 clinical trials. A synergistic evolution of MAbs and protein A resins also has taken place in the market. Figure 1 shows a graph adapted from an Amgen study of the development of protein A productivity and capacity over the past 40 years. Protein A has made MAbs highly manufacturable and easier to develop in laboratories. The purification platform approach, which has been enabled by protein A, has been a key element in this market growth.
As Figure 1 shows, productivity of protein A resins has increased >4% annually since 1978 and capacity about 6% annually. Despite that, opportunities exist for improving protein A technology. Upstream titers have improved dramatically, increasing nearly 100-fold over the past four decades. As such, protein A has become the rate-limiting step in some downstream purification suites.
In general, protein A columns are too large, which means that they either limit the use of prepacked technology, or they create a mismatch between a protein A column and downstream operations. Protein A columns also are more prone to bioburden contamination than any other step in a downstream process. Protein A resin is a reused raw material. It’s exposed to the highest load of nutrients as the entire harvest from the bioreactor is loaded onto the protein A column, but it is cleaned with weaker clean-in-place (CIP) chemicals than other downstream steps. Typically, ion-exchange and hydrophobic interaction chromatography (HIC) resins are cleaned with 1.0 M sodium hydroxide. But in a vast majority of cases today, protein A resin is cleaned with 0.1 M sodium hydroxide. At the same time, regulatory agencies are increasingly interested in sources of bioburden and the methods that manufacturers are using to limit the outbreak of bioburden in their manufacturing areas. And despite generational improvements that have led to lower cost of ownership for protein A, further cost reductions are needed.
Improving Process Productivity
Some challenges can be addressed through a simple process design. Several years ago, GE Healthcare introduced a technique called “variable residence time (VRT) loading.” VRT loading is a way to better use the capacity available in a resin while minimizing the amount of time needed during the load step. The technique is quite simple: Rather than loading a column at a constant residence time, you load a column in a step-wise manner. You start with a short residence time, and as the resin starts to reach a saturation point, you slow down the feed pump. Then you continue to load until reaching the saturation point and then slow down again.
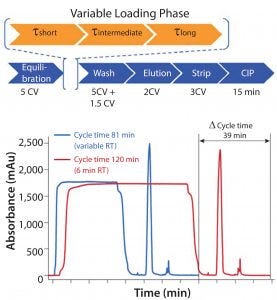
Figure 2: Variable residence time (VRT) loading
Figure 2 shows three different residence times for the load. The process leads to a shorter average residence time overall but maintains the capacity you would get with the longest residence time. Because the load step tends to be time-consuming during protein A chromatography, you can get significantly improved productivity. In this case, we demonstrated about a 40% productivity increase that maintains capacity and purification performance.
Another way protein A productivity can be improved is by implementing continuous capture, which is an area of focus. Continuous capture improves use of the available capacity in a resin, which allows the use of smaller equipment and in many cases, shorter process times and less manual handling. Continuous chromatography also makes it possible to manufacture smaller batches, which can be scaled-out instead of scaled-up.
Finally, fewer purification steps can be used in a process to improve overall productivity. For example, we have worked with customers to look for options for combining separate ion-exchange and HIC steps into a single multimodal step that provides the same purification performance but reduces a column in the downstream manufacturing area.
Improving Resin Productivity
Resin performance also has improved recently. To develop a next-generation version of protein A resin, we started by going back to MabSelect SuRe ligand, which uses a single domain built into a tetramer. We went back to the monomeric version and identified weak points in the protein A molecule to identify possible mutations that would improve alkaline stability. We used a high-throughput screening approach.
We also looked at the beads that we use for our protein A resins and thought a lot about the challenges the MabSelect SuRe bead was designed to address. MabSelect SuRe was developed at a time when many processes were at relatively low titers, and flow rate in the protein A step was one of the main limitations. Today most processes are at relatively high concentrations and, in fact, mass throughput is the limitation in the process, rather than volumetric throughput.
We considered opportunities to optimize a new bead for protein A chromatography, and we decided that it would be opportunistic to cooptimize both the ligand and the bead together. We performed experiments to cooptimize pore size and ligand length. Large pores reduce steric hindrances, but at the expense of available surface area. Long ligands increase the number of binding sites but can increase steric hindrances. As a result of our experiments, we found that there was an optimal design that maximized capacity while limiting steric hindrance.
The result is a new generation of protein A resin named MabSelect PrismA. MabSelect PrismA is built on the heritage of former MabSelect products, but it is a completely new bead and new ligand designed for modern processes. It offers improved binding capacity to resolve these bottlenecks that occur in production, and exceptional alkaline stability for cleaning and sanitization operations. And it has been designed from the start with an extensive security of supply package in mind.
Resin Performance
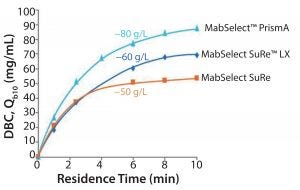
Figure 3: Resin performance, dynamic binding capacity (DBC)
Dynamic Binding Capacity: Figure 3 shows a comparison of MabSelect PrismA, MabSelect SuRe, and MabSelect SuRe LX resins tested at different residence times and measuring dynamic binding capacity (DBC). With MabSelect PrismA, the cooptimization of the ligand and base matrix has shown good improvement in terms of DBC at all residence times. Regardless of whether you run a continuous process in which residence times tend to be short, or you’re running a large-scale batch process in which they tend to be longer, you get a significant improvement of 25-40% improved capacity over MabSelect SuRe LX. MabSelect PrismA has a capacity that is on par with modern ion-exchange resins.
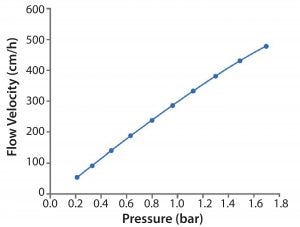
Figure 4: Flow rate from a process-scale column packed with MabSelect PrismA
Flow Rate: The new bead has been developed to meet the needs of today’s processes in terms of flow rate. Figure 4 shows data from a process-scale column packed with MabSelect PrismA. Both back pressure and flow rate were measured to determine the relationship between them. MabSelect PrismA offers very high flow rates at relatively low back pressure and does this with a bead approximately 30% smaller than MabSelect SuRe.
The combination of this higher capacity and optimized flow rate performance offers significant improvements in terms of productivity from existing equipment. Improved productivity means that if your plant is in a situation in which you need to make capital investments to expand, you might be able to delay those investments by making a change to MabSelect PrismA.
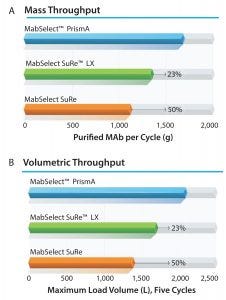
Figure 5: Throughput of a 450/200 column when loaded with a 4 g/L MAb feed
Productivity: Figure 5a shows a comparison of the mass throughput on a 45-cm column using MabSelect PrismA, MabSelect SuRe, and MabSelect SuRe LX resins. Mass throughput increases on the order of 25-50% depending on the benchmark. A similar relationship is true for volumetric throughput and the maximum-sized bioreactor that could be harvested with that column (Figure 5b). Using a 45-cm column, MabSelect PrismA could harvest a 2,000 L bioreactor with a titer of 4 g/L, whereas MabSelect SuRe LX and MabSelect SuRe resins would require larger columns for the same bioreactor.
Reduced Cost: The performance of MabSelect PrismA also can reduce the cost of consumables. Figure 6 shows the advantages in both resin consumption savings and buffer consumption savings compared over different resins. It shows an improvement between 20% and 33% in both resin and buffer consumption.
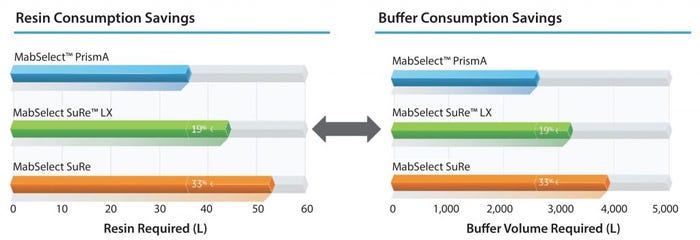
Figure 6: Raw material requirements to purify a 2,000 L load at 4 g/L
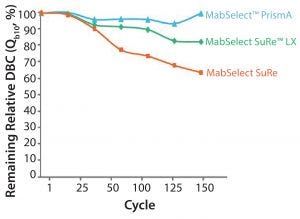
Figure 7: Cleaning with 0.5 M NaOH, 15 min contact time per cycle
Alkaline Stability and Bioburden Control: MabSelect PrismA offers significantly improved alkaline stability over other resins. Figure 7 shows a comparison of MabSelect PrismA against MabSelect SuRe LX and MabSelect SuRe resins when cleaned with 0.5 M sodium hydroxide. MabSelect PrismA retains more than 95% of its DBC after 150 cycles, whereas MabSelect SuRe LX has lost nearly 20% of its binding capacity and MabSelect SuRe has lost nearly 40%. So when designing processes, the capacity that you measure at cycle zero is still available even after repeated usage as compared with MabSelect SuRe LX or MabSelect SuRe resins. Another way to think about it is that safety factors can be reduced, thereby shrinking the size of your columns.
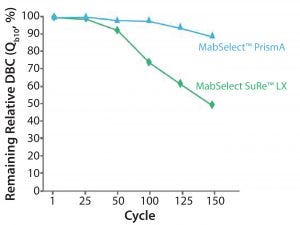
Figure 8: Cleaning with 1.0 M NaOH 15 min contact time per cycle.
If improved cleanability or resin cleanliness is a goal, you can increase sodium hydroxide concentrations to 1.0 M. Figure 8 shows a comparison of MabSelect PrismA and MabSelect SuRe LX resins cleaned with 1.0 M sodium hydroxide. MabSelect PrismA resin retains more than 90% of its dynamic binding capacity, whereas MabSelect SuRe LX retains only about 50%. This is an important improvement in terms of performance if you want to align your CIP solutions throughout the entire downstream suite or if you want to have better control of bioburden.
Purification Performance: We have conducted a study using two of our in-house MAbs to compare the purification performance of MabSelect PrismA, MabSelect SuRe, and MabSelect SuRe LX. The load values are quite different: MabSelect PrismA has been loaded with about 50% more MAb due to its higher dynamic binding capacity.
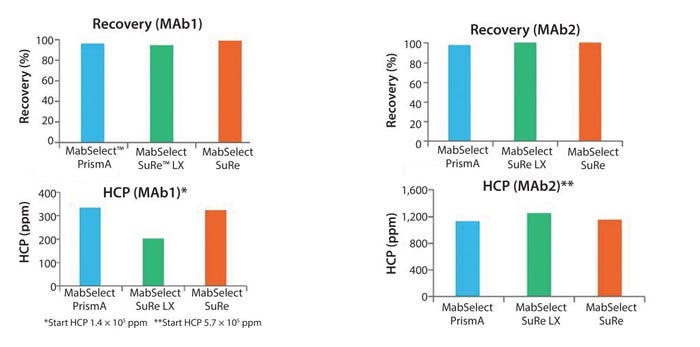
Figure 9: Recovery and host-cell protein removal
The top of Figure 9 shows the two MAbs tested and a comparison of recovery. In all three cases, we had very similar performances. Host-cell protein levels are shown in the eluate pool at the bottom of Figure 9, which again shows equivalent performance between the three resins.
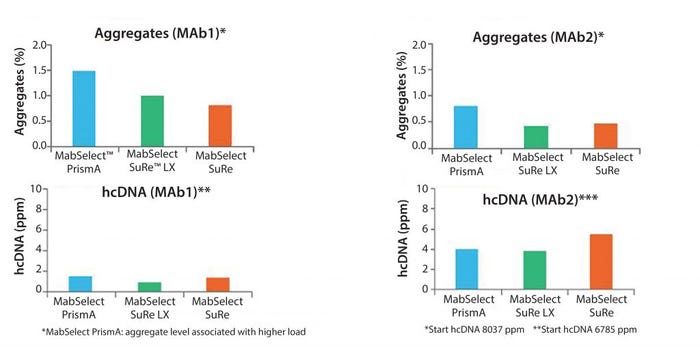
Figure 10: Aggregate and host-cell DNA removal
The top of Figure 10 shows a comparison of aggregates. The bottom of this figure shows DNA clearance on the three resins. Purification performance is very similar.
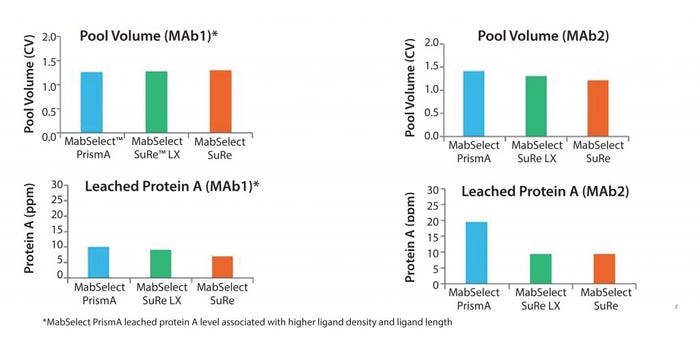
Figure 11: Elution pool volumes and leached ligand content
Figure 11 shows pool volumes and leached protein A. Pool volumes are similar across the three resins. For leached protein A, there are slightly higher values of protein A for the resins that have higher ligand densities. This is expected, but in all cases we believe all of these values are easily handled by the secondary and tertiary steps in a downstream purification suite.
GE Healthcare has put extended measures into MabSelect PrismA to ensure strong security of supply. From the start, we’ve designed this product to have two sources of agarose validating into the manufacturing and two sources of the protein A ligand. We also have taken steps to develop in-house manufacturing for the protein A ligand. As a second source, we have a contract manufacturer that manufactures the ligand using a GE-developed process.
Conclusion
Productivity can be improved in MAb downstream processing, by improving both process designs and resins. MabSelect PrismA has been developed to address some of the challenges we have today. It’s a new high-capacity resin with resistance to 1.0 M sodium hydroxide for solving both productivity and bioburden issues. It offers significantly higher binding capacity, which gives improved mass throughput and productivity, especially when looking at existing equipment and trying to get the most out of your existing capital.
Learn more: 
Jonathan Royce is Global Business Leader for Chromatography Resins at GE Healthcare Life Sciences.
You May Also Like





