It is no big revelation that publishing is changing. How people prefer to read and access information is in some ways a demographic conundrum and in other ways an expected technology-driven evolution. Recognizing those changing attitudes and in honor of our 15th anniversary year as a publication, for 2017 BPI is planning to both adapt to and further drive business and publishing trends in ways that truly meet the information needs of our readers around the world. We simply didn’t have many of these options 15 years ago!
Here are a few changes in the works — and how they align with industry perspectives:
As I mentioned last month, prospective authors can access our new guidelines online — www.bioprocessintl.com/about/author-guidelines — which include more specific information about graphics preparation and house style (with the final list of our upcoming themes and subthemes).
This issue’s report on our 2016 BPI Awards results also reflects the importance of new industry mind-sets. The nominations for coll...
BPI Advisor Will Be Missed
Thomas J. Pritchett
, PhD (1950– 2016), was a member of the
BioProcess International
advisor board since its founding. He also was founder, publisher, and editor of the
BioQuality Newsletter
and provided decades of analytical and quality control training to many people in the biopharmaceutical industry, both on the industrial and regulatory sides. Pritchett always provided insightful, well–thoughtout approaches to implementing current good manufacturing practices (CGMPs) under all conditions, ranging from virtual to global companies. He helped to train emerging regulatory systems in the Middle East. His focus was always on patient safety and good science. Tom will be dearly missed by those of us who had the pleasure of working with him.
Pritchett committed suicide after many years of struggling with bipolar disorder. It is his family’s request that those of us in the biopharmaceutical industry lead the way to understanding that mental diseases are illnesses that need our inn...
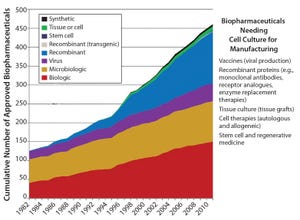
Figure 1: The number of cell culture-based products is growing
What prompts over 100 biopharmaceutical manufacturers, leading academics, supply partner R&D heads, regulators, and worldwide regional hubs to get involved in a major project? It’s when that project identifies the future technology needs of the biopharmaceutical manufacturing industry and accelerates its collective innovation. In February 2015, the BioPhorum Operations Group (BPOG) Technology Roadmapping steering committee met in Washington DC to create the first “technology roadmap” for the biopharmaceutical manufacturing industry.
The biopharmaceutical market has been experiencing dramatic changes: explosive growth coupled with increasing pressures to reduce costs while producing effective drug products. Such changes have forced manufacturers to change their legacy processes. At the same time, innovation has been slow in the bioprocess industry, with biomanufacturers developing technology in isolation and suppliers unclear on what biomanufac...
India’s position as a global participant in small-molecule generic drugs, vaccines, and enzymes has been proven over decades. The country is one of the most populous and fastest-growing regions in the world, both economically and technically. But India’s potential as a biologics participant has not been realized. Its competence as a global biologics producer has not yet caught up.
Global industry concerns regarding the country’s position in the (bio) pharmaceutical industry haven’t changed much over the past eight years since my company completed its study,
Advances in Biopharmaceutical Technology in India
(
1
). Issues include political volatility, a desire for increased spending on domestic healthcare, intellectual property protection, and a need to develop India’s existing talent and education systems to meet the needs of biopharmaceutical technology segments. And India’s struggle to fully invest in innovative research and development (R&D) may be creating long-term gaps in the country’s progress.
Ca...
WWW.GETTYIMAGES.COM
As a cofounder of Wave Biotech (now a division of GE Healthcare), my partners and I often struggled with critical choices regarding partnering and funding opportunities. Every new, attractive, and potentially disruptive technology will court attention once it experiences some modest adoption and acceptance, even while attempting to “fly under the radar” of major players. The challenge for life-science entrepreneurs is how best to navigate those decisions and select the right path as company founders. Weighing and evaluating potential partners and funding sources is a common cause of stress among our entrepreneurial colleagues.
In our case at Wave, we debated whether we were ready to work hand in hand with a venture capital (VC) partner. In the early 2000s, there was a lack of VC and private equity (PE) investors possessing a deep understanding of the life-sciences tools and services (LSTS) sector. With nearly all potential VC partners considered, we would have been one of the few LSTS ...
BPI publisher, Brian Caine
Since 2003, the mission of
BioProcess International
has been to connect biopharmaceutical scientists and decision makers to the science, technology, and expertise that can positively influence and improve existing bioprocesses. Our
BioProcess International
Awards were created in 2012 to mark the magazine’s 10-year anniversary. They allow us to reflect on and help honor the time and investment companies put into researching, developing, and launching biopharmaceutical products, technologies, and services to deliver better, more efficient treatments and increased hope to a global patient base.
To date, these biennial awards have recognized more than 50 winners who represent excellence in leadership, corporate citizenship and collaborations, innovative facility design, emerging companies, and best application of technologies in upstream and downstream processing and analytical methods. The BPI Awards also singles out individuals who personally challenge the biopharmaceutical in...
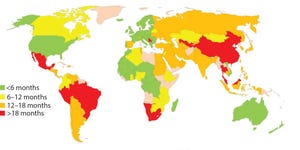
Figure 1: Estimated global approval times for major changes based on industry experience (e.g., new drug product manufacturing site)
Pharmaceutical products save or improve the lives of millions of people each year. Thorough regulatory review of chemistry, manufacturing, and controls (CMC) information is critical to ensure drug product safety, quality, and efficacy as well as to secure patients’ continuous access to such products. But achieving all of that at an effective cost is difficult. Companies race to launch products to patients as soon as possible after clinical efficacy is demonstrated.
Biomanufacturers often need to make changes such as increasing batch sizes, adding new manufacturing facilities to expand patient access, improving analytical methods, and increasing process robustness as companies gain experience in commercial manufacture and testing. Changes also may be made to comply with new regulatory expectations. But before changes are made, manufactures must evaluate process risks and ofte...
Photo 1: A 30-cm OPUS column containing CaptivA resin connected to an ÄKTA Ready skid (GE Healthcare) for a good manufacturing practice (GMP) operation
In downstream purification of monoclonal antibodies (MAbs), the single greatest contributor to manufacturing costs is the expensive capture step typically based on protein A affinity chromatography. Almost since its introduction to bioprocessing, efforts have been made to reduce the cost of this step.
Several alternative ligands have been promulgated as potential replacements for protein A, but they have proven difficult to adopt and scale up. Supplier companies have pushed for increases in capacity and economics, but those are always accompanied by a corresponding increase in purchase price. Although increased capacity, higher flow rates, and increased caustic and process stability all have their price, Repligen believes that in most cases at clinical-phase manufacturing, little or no economic benefit comes from the extended cleanability or capacity that ...
Serena Fries Smith, associate director of global technical engagements for Thermo Fisher Scientific, covered advantages in feed solutions in the context of current industry trends in an early November webinar. Leveraging feed designs and strategies can optimize glycosylation of complex proteins, simplify scale-up of fed-batch processes, and improve expression titers.
Smith’s Presentation
In 1994, the average expression titer was 0.5 g/L. Thanks in part to improvements in culture feeding, titers had increased to 1.3 g/L in 2004. By 2014, they reached 2.5 g/L. Now some processes are reporting up to 7.0 g/L. And Thermo Fisher has begun to tackle other industry challenges: e.g., developing feed formulations that help modulate glycosylation (GlycanTune total feeds) and dry media for simplified handling and improved process scale-up.
Glycosylation is an important protein quality attribute. It is critically important when a glycan distribution profile must be matched (e.g., when developing biosimilars or transit...
Orjana Terova is a purification product manager in the bioproduction division of Thermo Fisher Scientific. In a BPI webinar on 9 December 2016, she discussed the company’s custom resin program for purification of biological products. Thermo Fisher Scientific has dedicated a pilot-plant facility for this program.
Terova’s Presentation
Speed is the main development driver in downstream processing, but quality and efficiency are always critical. Purification processes need the highest resolution, capacity, salt tolerance, and operation speed possible. Consistency and reproducibility are important, as are cost minimization and mitigation of supply risk.
Adenoassociated viruses (AAVs) have emerged as the vectors of choice for many gene therapies because they can mediate long-term, tissue-specific gene expression with low immunogenicity. The biopharmaceutical industry can purify viral vectors on a scale for clinical supply, Terova said, but it needs industrialized platform technologies to maximize productivity ...
Sponsored Content
In an early October webcast, Surendra Balekai (senior global product manager for bioproduction at Thermo Scientific) discussed improving bioreactor design for a 5:1 turn-down ratio. Balekai has worked for over 17 years with biological manufacturers in designing processing equipment for developing vaccines, blood products, and recombinant proteins.
Balekai’s Presentation
When bioreactors operate at a 5:1 ratio (20% volume), the primary challenges are carbon dioxide build-up in the headspace and fluid mixing at low volume. The CO
2
produced will significantly affect the cell culture process by reacting to create an acidic environment that affects cell growth and viability. Dynamically in a low-volume system, the positions of the impeller, spargers, and probes, are all as important as temperature control. The impeller could be oversized and create a relatively larger shear zone at low volumes. If the sparger is in the wrong position, it won’t be able to flush gases out effectively. To provide effective heat...
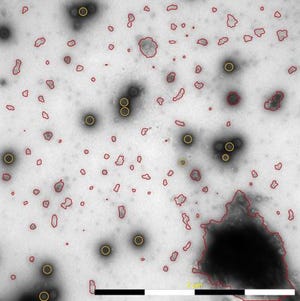
In an October webcast, Vironova’s Josefina Nilsson (head of electron microscopy services) and Gustaf Kylberg (MiniTEM product manager) discussed subvisible particle characterization using the MiniTEM system, which provides both morphology and quantitative data.
Nilsson and Kylberg’s Presentation
Vironova combines expertise in electron microscopy and image analysis with in-house designed software. Electron microscopy is a good technique for getting to know your product in detail (morphology and other characteristics). After discussions with partners and clients, Vironova worked with Delong Instruments to develop the tabletop MiniTEM transmission electron microscope.
Undesired outcomes can occur during biopharmaceutical development, manufacturing, and storage: e.g., aggregate formation, failure to remove host cell debris, and loss of particle morphology or integrity. Electron microscopy can show you whether such outcomes are occurring.
By contrast with a conventional electron microscope that requires a high...