June 2019 Featured Report
Photo 1:
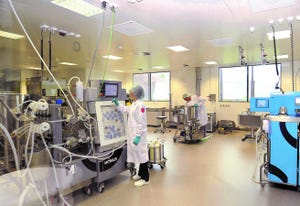
Large-scale downstream area at Merck Serono in Vevey, Switzerland (WWW.EMDGROUP.COM)
Early manufacturing facilities for large-scale production of biopharmaceuticals were, by necessity, very large. Low expression titers and blockbuster-market products such as monoclonal antibodies (MAbs) combined to require massive bioreactors — with capacities of 10,000– 20,000 L or more — and supporting infrastructure. In my early days covering the industry, I visited a few such facilities and was always awed by the huge tanks and what seemed like miles of piping. A 2010
Pharmaceutical Engineering
article described a process modeling approach to building a traditional large-scale facility for Merck Serono in Vevey, Switzerland (Photo 1):
Photo 2:
On the cover, the 2005 ISPE award-winning modular facility designed by NNE for Novo Nordisk was a “fast-track” 18-month project (WWW.NNE.COM).
That epitomizes what may be considered as the apex of multiproduct facility design based on stainless-steel reusable equipm...
(WWW.PALLBIOTECH.COM)
The BioPhorum first-edition Technology Roadmap outlined a 10-year vision for therapeutic protein production in the biopharmaceutical industry (
1
). The roadmap describes multiple manufacturing scenarios ranging from large-scale (~20,000-L production) to small and agile, portable production facilities. It includes detailed analyses of the needs for the future in each of the following areas:
Since the 2017 publication of the technology roadmap, the BioPhorum Technology Roadmapping Phorum has prioritized key needs outlined in it and put into place a series of follow-on activities designed to accelerate the technology development that the roadmap describes. At the same time, further detail is being added to the roadmap documents, and a vision of the future continues to be developed. The next analysis on continuous bioprocessing will be published soon, with particular emphasis on downstream manufacturing.
Continuous bioprocessing
has been proposed as one future biomanufacturing state be...
Drug Substance Scenarios
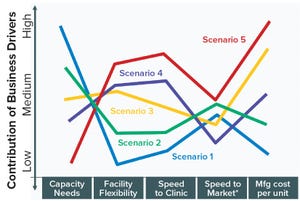
Given the complexity of the biopharmaceutical industry and the increasing diversity of products and companies, it is clear that there will be no “one size fits all” solution to biomanufacturing. Instead, we see a range of biomanufacturing scenarios playing out over the next 10 years. Five high-level scenarios were selected for drug substance manufacturing and two for drug product to cover the full spectrum of process and facility types. Each facility type is associated with a representative set of business conditions reflecting a typical mix of key drivers and business dimensions.
Table 1:
Main drug substance facility scenarios with typically associated product types and business drivers
Table 1 summarizes the drug substance scenarios and includes a list of characteristic products and facility features. More detail on each facility type/ scenario follows in the subsections below to provide a vision from which enabling technologies were identified. Since the relative value or impa...
In past decades, the focus in bioprocess engineering was on traditional stainless steel project management, which is highly dependent on established project schedules. Many milestones, tasks, and deliverables during basic and detailed design and execution were implemented in the same tried and tested way by engineering, suppliers, and biopharmaceutical companies. Making decisions was complicated by alignment with long lead items. With the introduction of single-use (SU) technologies, the transition in execution and optimization of project management only just has been fully understood. When SU technologies were first introduced, many experienced and established engineering companies tried to reuse their proven workflows for them. Now they recognize that the technical innovations also require a rethinking of project execution.
Today, there is great demand for adapting traditional bioprocess project management activities. Customers now specifically need flexible facility layouts and platform processing incl...