Container Materials for Biopharmaceuticals: A Comparative Small-Scale Case Study of Stainless Steel and a Proprietary Nickel-Based AlloyContainer Materials for Biopharmaceuticals: A Comparative Small-Scale Case Study of Stainless Steel and a Proprietary Nickel-Based Alloy
Evaluating compatibility of a drug substance with all surfaces that it might come into contact with during drug product manufacturing is essential to ensure product quality. Proteins can adsorb to contact surfaces, form aggregates, and desorb into a drug-substance solution. Proteins also can degrade in presence of leachables generated from contact surfaces during manufacturing.
Containers and vessels used during manufacturing are single-use disposable components or metal tanks, primarily either 316L stainless steel (SS) or C-276 Hastelloy nickel-based alloy (HLY). Researchers have shown protein degradation in presence of metal contact surfaces. In two separate studies, Bee et al. (1) and Kalonia et al. (2) reported adsorption of monoclonal antibodies (MAbs) on SS surfaces and subsequent aggregation, attributing the phenomenon to structural alteration or unfolding caused by the adsorption step. Different formulation factors (e.g., buffer species, pH, volume per unit contact surface area, protein concentration, contact time, and metal chelator concentration) have been shown to have influence metal leachables because of contact with SS over time (3, 4).
The origin of metal leachables, their impact on protein product quality and safety, and the analytical tools to identify and measure them quantitatively have been discussed (5). Testing of the impact of metal ions on product quality as part of formulation and process development also has been proposed (5).
Lam et al. reported on the oxidation of recombinant humanized monoclonal antibody (rhuMAb) human epidermal growth factor receptor 2 (HER2) (6). In that study, a formulation containing sodium chloride (NaCl) was filled using an SS filler and subsequently stored at 40 °C for two weeks. The oxidation was attributed to corrosion of SS by NaCl and was probably generated by the iron ions in the formulation exposed to SS.
Chen et al. reported on aggregation of a MAb (7) after it had undergone bulk freeze–thaw in a stainless-steel vessel and subsequent storage at 40 °C over time. Another group of researchers evaluated the impact of stainless-steel exposure by storing protein samples in stainless-steel cups and glass vials over two weeks at 25 °C and 40 °C, subsequently aliquoting samples into glass vials and monitoring their stability over six months at 2–8 °C and 25 °C (8). Increased aggregation and oxidation were observed after samples were stored in glass vials for six months at 25 °C, which correlated with the initial length of exposure in SS cups (8). Klair et al. reported oxidation-induced cross-linking in MAbs during a stress temperature (40 °C) study in small-scale HLY containers and attributed that observation to metal leaching (9).
We compared HLY (a nickel-based alloy) with SS (an iron-based alloy) and for compatibility with two MAbs. Holding the drug substance in both types of metal containers at ambient conditions — 25 °C and 60% relative humidity (RH) — for 14 days simulated the worst case in process manufacturing hold time. We then aliquoted the contents of the metal containers into glass vials to assess subsequent changes in quality attributes during thermal stress at 40 °C and 75% RH. Our findings provide key insights on a direct comparison between SS and HLY as metal containers used for drug-product manufacturing.
Materials and Methods
Materials: We formulated IgG1 MAbs in a histidine buffer containing sucrose and polysorbate 80 at pH 6.0 at a protein concentration of 120 mg/mL. We used 25-mL passivated and electropolished 316L SS and C-276 HLY miniature tanks with an internal diameter of 3.48 cm. Before using those tanks, we visually examined their surfaces to confirm the absence of cracks, scratches, corrosion, and other deformities. We washed and autoclaved the tanks before use.
Ambient Hold: We filled, washed, and autoclaved 316 L SS and C-276 HLY miniature tanks with 21 mL each of two MAb drug substances (MAb A and MAb B). We held the tanks at 25 °C and 60% RH inside a controlled temperature and humidity chamber with no lights (ES2000 CDM-BT, Bahnson Environmental Specialties LLC) over 14 days. We achieved a surface-area-to-volume ratio of 1.45 cm–1 using a fill volume of 21 mL. We filled three miniature tanks for each tank type and MAb to capture all intertank variabilities. Drug substance filled in type I glass vials (3 mL in 6-mL glass vials) was held under the same conditions and served as the control. We pulled 3-mL aliquots from each vessel at 0, 1, 2, 4, 7, 11, and 14 days. Those samples were tested for soluble aggregates using size-exclusion–ultraperformance liquid chromatography (SE-UPLC), for charge variants using capillary isoelectric focusing (iCIEF), for subvisible particulate counts using micro-flow imaging (MFI, ProteinSimple MFI 5200), for visible particulate matter using manual inspection, and for pH (using a Seven Excellence Mettler Toledo pH meter and pH Module S400).
Variable-pathlength spectroscopy, SoloVPE (Repligen Agilent Cary 60 UV-vis) provided a measurement of MAb concentration. We visually examined the general appearance of the samples, monitored oxidation using reduced peptide mapping with liquid chromatography–mass spectrometry (LC-MS), and measured trace metal levels using inductively coupled plasma mass spectrometry (ICP-MS).
Thermal Stability: For the ambient hold study, we washed and autoclaved 316 L SS and C-276 HLY miniature tanks, filled them with 21 mL each of the two MAb drug substances, and held them at 25 °C and 60% RH for over 14 days. At day-7 and day-14 of the ambient hold, we pulled aliquots from both tank types and poured them into 6-mL type I glass vials in preparation for the thermal stability set down (Figure 1).
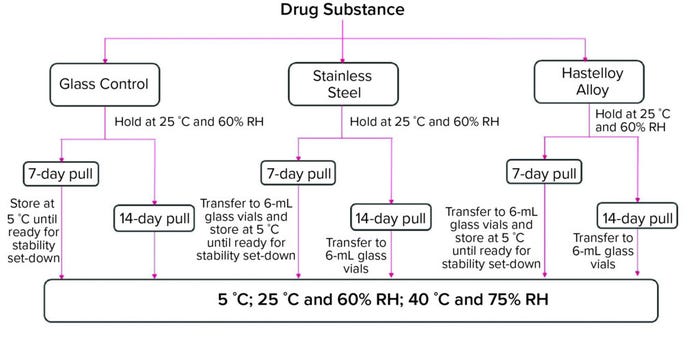
Figure 1: Thermal stability study after seven- and 14-day ambient holds in glass, stainlesssteel,
and Hastelloy alloy containers; analytical testing measured soluble aggregates (size
exclusion–ultraperformance liquid chromatography, SE-UPLC), charge variants (imaged
capillary isoelectric focusing, iCIEF), subvisible particulate counts (microflow imaging, MFI),
general appearance, protein concentration (C Technologies, Inc. Agilent Cary 60 UV–vis),
oxidation (liquid chromatography–mass spectrometry, LC-MS). (RH = relative humidity).
The aliquots and the glass-vial controls were set down on stability at 5 °C, 25 °C and 60% RH, and 40 °C and 75% RH conditions. Stability samples underwent testing for all product quality attributes as listed in Figure 1 except for oxidation using reduced peptide mapping. We conducted that test only on day-14 samples pulled at one- and two-month time points from the 40 °C and 75% RH condition, the three-month time point from 25 °C and 60% RH condition, and the three- and six-month time points from the 5 °C condition. We also pulled day-7 samples at the six-month time point from the 5 °C condition group and tested those for oxidation. Only the 40 °C and 75% RH oxidation data are presented herein.
Size-Exclusion Chromatography: We used a Waters Acquity UPLC system and an Acuity BEH 4.6-mm × 300-mm SE column for SE-UPLC analysis. We loaded 36 µg of nominal protein amount onto the column and quantified protein using 280 nm as the absorbance wavelength. The system ran in isocratic mode using 10-mM phosphate buffer with 500-mM sodium perchlorate at pH 7 as the mobile phase buffer at a flow rate of 0.3 mL/min.
Peptide Mapping Using LC-MS: LC-MS-based peptide mapping determined site-specific oxidation-level quantification. We denatured and reduced the protein samples in solution containing 5 mM of acetic acid and 5 mM of tris(2-carboxyethyl) phosphine hydrochloride by heating to 70 °C for 10 min. We diluted samples in 100-mM tris buffer (pH 8.0). Iodoacetamide and trypsin as an enzyme-to-substrate ratio of 1:20 (w/w) were added into samples, followed by incubation in the dark at 30 °C for 180 minutes. Addition of 1-mM urea and 0.5% trifluoroacetic acid stopped trypsin digestion.
We performed LC-MS analysis using a Waters Acquity UPLC system coupled to a Q Exactive Plus Hybrid Quadrupole-Orbitrap mass spectrometer (from Thermo Fisher Scientific). Peptides were separated in a BEH C18 column (1.7 µm, 2.1 mm × 150 mm) with a gradient of 0.1–40% acetonitrile with 0.5% trifluoracetic acid over 90 minutes at a flow rate of 0.25 mL/min. A photodiode array detector monitored eluted peptides at a wavelength of 215 nm before MS data acquisition. Byonic software (Protein Metrics Inc.) processed the LC-MS data, which we used to generate peptide maps and identify oxidation sites. We quantified relative levels of oxidation from each site based on extracted ion chromatograms using Skyline-daily software.
Trace-Metal Analysis with ICP-MS: We used a Perkin Elmer 300 series ICP-MS instrument to quantify samples for metal content after diluting them in 0.2% nitric acid/0.1% Triton X surfactant. We aspirated the samples into ICP-MS and extrapolated concentration of elements in the sample from a two-point calibration curve made from blank and standard injections and from predefined instrument-response factors.
Charge Variants Analysis with cIEF: We analyzed charge variants using isoelectric focusing (ProteinSimple iCE3 system) and a fluorocarbon-coated cIEF cartridge. We diluted samples to 2.5 mg/mL in Milli-Q water and subsequently diluted them to 0.5 mg/mL to yield final sample solution of 0.5 mg/mL protein, 2% Pharmalyte 3-10 carrier ampholytes (from Cytiva), and 2% Pharmalytes 8-10.5 carrier ampholytes, 2 M urea, 0.35% methyl cellulose, and isoelectric point (pI) markers (6.61 and 9.77, from ProteinSimple).
Results and Discussion
Ambient Hold: For both MAbs and all containers, visual appearance and pH did not change during ambient hold over 14 days. High–molecular-weight percentage (%HMW) levels showed a statistically significant change over 14 days ambient hold (Figure 2) for MAb A and MAb B in all three container types. A pairwise comparison of day-14 %HMW levels performed using Tukey’s honestly significant difference (HSD) test showed significant difference (p < 0.05) between glass and SS for both MAbs (Table 1). A minor difference was observed in day-14 levels between container types for subvisible particle counts in the 2–10-µm range (Table 1). Glass containers accounted for a slightly higher count than containers made of HLY and those made of SS. However, that difference can be attributed to method variability.
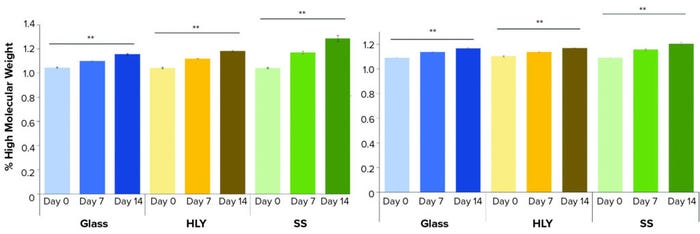
Figure 2: Percentage of high–molecular-weight over 14 days hold at ambient conditions for (left) MAb A and (right) MAb B; levels at day-0 and day-14 were compared using Student’s t-test. (** means p-value is <0.01, SS = stainless steel, HLY = Hastelloy alloy).
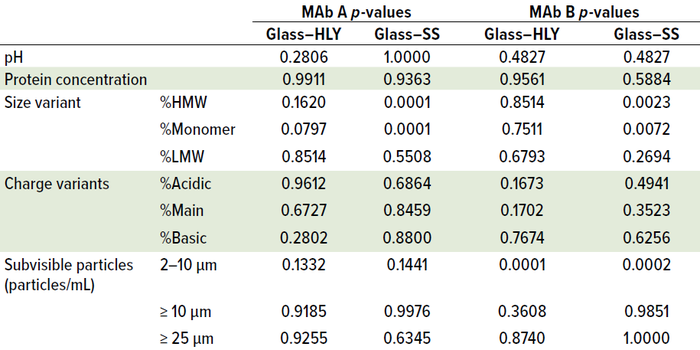
Table 1: Day-14 comparison in quality attributes using Tukey’s honestly significant
difference (HSD) test across container types after 14 days of ambient exposure; a pairwise
comparison showed significant difference between glass and stainless steel (SS) %HMW
and %monomer for both monoclonal antibodies (MAbs) (p < 0.05). (HLY = Hastelloy alloy).
Peptide mapping analysis with LC-MS showed that oxidation levels at site M255 for HLY and SS as well as site M431 for SS containers of MAb A increased with holding time. M104 for glass and HLY, and site M255 for HLY and SS with MAb B slightly increased as the holding time increased in all containers (Figure 3). Changes over time also were statistically significant.
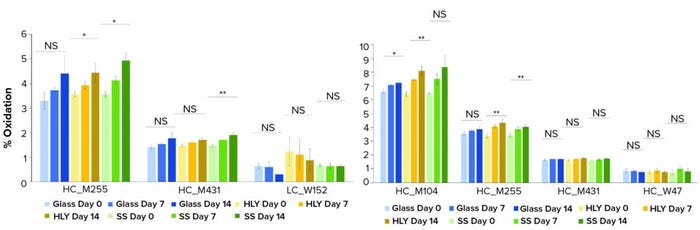
Figure 3: Methionine and tryptophan sites with levels above 1% oxidation for (left) MAb A and (right) MAb B over a 14-day hold at ambient conditions; levels at day-0 and day-14 were compared using Student’s t-test. (* means p-value is between 0.01 and 0.05; ** means p-value is <0.01, NS = not significant, SS = stainless steel, HLY = Hastelloy alloy).
Table 2 shows that no significant difference was apparent in oxidation levels between containers (p > 0.05) after we compared 14-day values using Tukey’s HSD test. No significant change was apparent for other post-translational modifications (PTMs) in all samples (data not shown). In line with results published by Lam et al. (6), elemental analysis showed consistent presence of iron and manganese in all SS samples. For MAb A, iron levels were 0, 255, and 367 parts per billion (ppb) in 0-, 7-, and 14-day samples, respectively. For MAb B, iron levels were 0, 235, and 283 ppb for those days. For MAb A, manganese levels were 0, 405, and 901 ppb for those days. For MAb B, manganese levels were 0, 384, and 920 ppb for those days. Those elements were not detected in either glass or HLY–exposed samples (Figure 4). It is important to note that despite the presence of those metal ions in samples exposed to SS, there was no immediate impact on methionine oxidation. However, absence of an immediate impact of leachables does not necessarily eliminate the possibility of degradation over long-term storage. Stressed thermal stability studies can provide useful insights into the potential impact of such leachables on product quality, as discussed below.
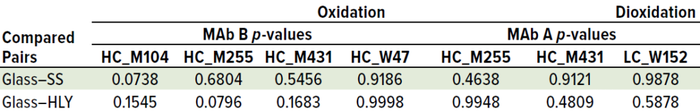
Table 2: Day-14 methionine and tryptophan oxidation level comparison using Tukey’s HSD test across container types after 14 days of ambient exposure; no significant difference was observed in oxidation levels between containers (p > 0.05).
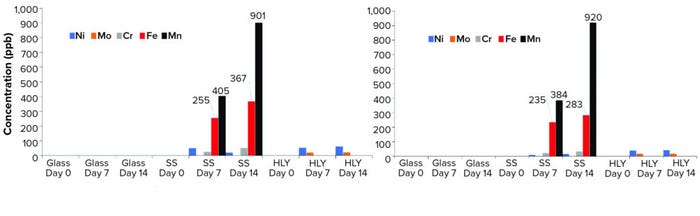
Figure 4: Trace-metal leachable profile of (left) MAb A and (right) MAb B; samples were exposed to ambient hold for 14 days; limit of quantification (LoQ) of 0.025 ppm applies to class 1, 2A, and 3 elements. LoQ for other elements is 100 ppb.
Thermal Stability: Both MAbs showed no visual change in appearance and had no change in pH for up to two months at 40 °C and 75% RH. The subvisible particle count in the 2- to 10-µm size range showed variability with no discernable trend, and no meaningful change was apparent in the ≥10-µm and ≥25-µm particle counts (data not shown). Protein concentrations also did not show meaningful change up to two months at 40°C and 75% RH (data not shown). Samples showed an increase in percentage of acidic species and a corresponding decrease in percentage of main and percentage of basic species observed with no observable effect of container type (Figure 5).
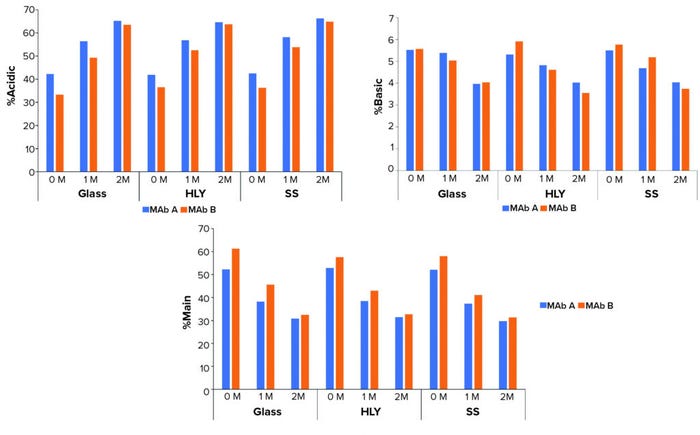
Figure 5: Effect of container type on percentage of charge variants at 40 °C and 75% RH; samples were staged on thermal stability for two months at 40 °C and 75% RH after exposure to glass, stainless steel (SS), and Hastelloy alloy (HLY) containers over 14 days in ambient conditions.
It is interesting to note, however, that peptide-mapping data showed that the levels of several quality attributes, including N-terminal pyroglutamate, methionine oxidation, aspartic acid cyclization/isomerization, and asparagine deamidation increased under thermal stress conditions across all three container types. The attributes that showed most significant changes over time at 40 °C and 75% RH were oxidation at site locations HC_M431 and HC_M255 and deamidation at HC_N328 and HC_N387/N392 for MAb A and for MAb B, oxidation at sites HC_M255 and HC_M104 and deamidation at sites HC_N328 and HC_N387/N392.
Most important is that methionine oxidation was affected considerably by type of container material. In SS containers, the most susceptible sites at HC_M255 and HC_M431 for MAb A and HC_M104 and HC_M255 for MAb B exhibited significant increases compared with glass and HLY containers in the samples exposed to 40 °C and 75% RH conditions (Figure 6, Tables 3 and 4).
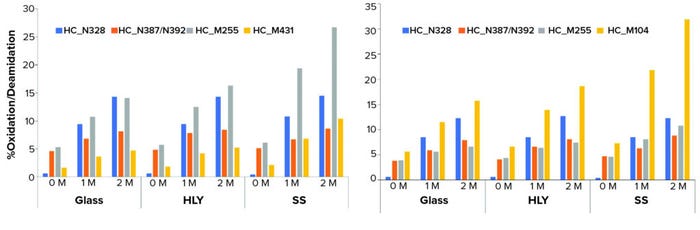
Figure 6: Effect of container type on methionine oxidation and asparagine deamidation at 40 °C and 75% RH for (left) MAb A and (right) MAb B; samples were staged on thermal stability for two months at 40 °C and 75% RH after being exposed to glass, stainless steel (SS), and Hastelloy alloy (HLY) containers over 14 days in ambient conditions.
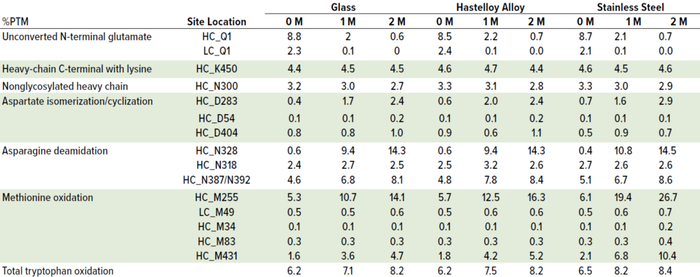
Table 3: Effect of container type on MAb A percentage of post-translational modification (%PTM) after 14 days of ambient hold; post-hold, samples were stored in glass vials at 40 °C and 75% RH over two months. (HC = heavy chain, LC = light chain).
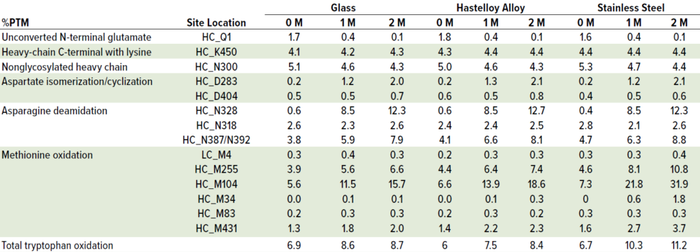
Table 4: Effect of container type on MAb B percentage of post-translational modification (%PTM) after 14 days of ambient hold; post-hold, samples were stored in glass vials at 40 °Cand 75% RH over two months. (HC = heavy chain, LC = light chain).
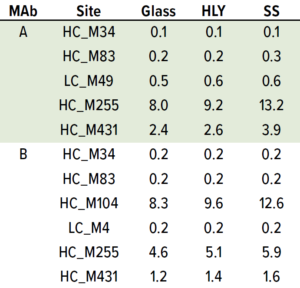
Table 5: Effect of container type on percentage of methionine oxidation at 25 °C and 60% RH; samples were staged on thermal stability for three months at 25 °C and 60% RH after being exposed to glass, stainless steel (SS), and Hastelloy alloy (HLY) containers over 14 days in ambient conditions.
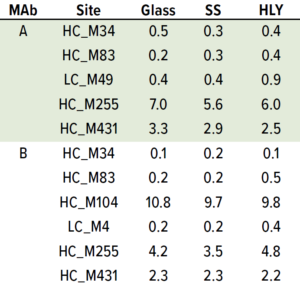
Table 6: Effect of container type on percent methionine oxidation at 5 °C; samples were staged on thermal stability for six months at 5 °C after being exposed to glass, stainless steel (SS), and Hastelloy alloy (HLY) containers over 14 days in ambient conditions.
Although M104 is located in the complementarity-determining region (CDR) of MAb B, potency was unchanged (data not shown). No other stress-susceptible PTMs such as asparagine deamidation, aspartic acid cyclization/ isomerization, and N-terminal pyroglutamate exhibited differential stability in different containers, even though they still showed time-dependent percentage increase (Tables 3 and 4). Level of total tryptophan oxidation/degradation products in MAb B were slightly higher in samples in SS containers than in glass containers under stress conditions (40 °C and 75% RH) conditions (Tables 4). Similarly, SS containers induced more oxidation than did glass and HLY containers at 25 °C and 60% RH observed at HC_M255 and HC_M431 for MAb A and HC_M104 and HC_M255 for MAb B (Table 5). There was no significant difference in oxidation level across container types at 5 °C for up to six months (Table 6).
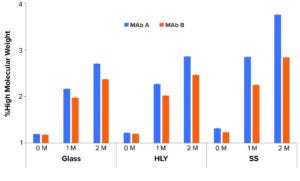
Figure 7: Effect of container type on percent high–molecular weightat 40 °C and 75% RH; samples were staged on thermal stability for two months at 40 °C and 75% RH after being exposed to glass, stainless steel (SS), and Hastelloy alloy (HLY) containers over 14 days in ambient conditions. A pairwise statistical comparison (Tukey’s honestly significant difference (HSD) test) of percent aggregates after two months showed significant difference (p < 0.05) between glass, HLY ,and SS containers for both MAbs.
Percentage of HMW levels for samples in SS containers trended higher than samples in glass and HLY containers at 40 °C and 75% RH (Figure 7). A pairwise statistical comparison (using Tukey’s HSD test) of aggregate percentages in day-14 samples after two months at 40 °C and 75% RH showed significant difference (p < 0.05) between glass, HLY, and SS containers for both MAbs. The difference in %HMW among glass control and samples in stainless-steel containers was higher than the difference between glass control and samples in HLY containers for both MAbs. The difference in %HMW among samples in glass and SS containers for MAb A was greater than the intraday standard deviation (Table 7).
Discussion
We demonstrated the relative inert nature of HLY compared with SS with respect to its effects on product quality (specifically, the impact on oxidation) of two MAbs. Our findings underscore the importance of testing the impact of thermal stress after initial ambient contact of a product with metal contact surfaces. Results showed no significant difference in product quality between HLY- and SS-exposed samples when held under ambient conditions for up to 14 days, except for a minor increase in %HMW. However, application of thermal stress on samples within glass vials after ambient hold in metal containers resulted in noticeably higher oxidation in SS-exposed samples. ICP-MS testing showed presence of iron and manganese in samples exposed to SS surfaces, indicating the possible role of metal leachables from SS containers on oxidation of the MAbs. Potency was unchanged for both MAbs used in this study.
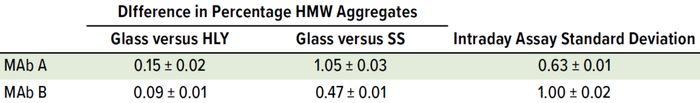
Table 7: Effect of container type on percentage of aggregates; samples were staged on thermal stability for two months at 40 °C and 75% RH after being exposed to glass, stainless steel (SS), and Hastelloy alloy (HLY) containers for 14 days under ambient conditions.
Our study directly compared metal types and assessed thermal stability postexposure of drug substance to different metal containers. We identified differences between metal types with respect to their potential impact on product quality. Such an approach for compatibility assessment can be useful in selecting metal containers or in strategies to limit overall exposure time to minimize impact on product quality. We plan to expand this work to include other MAbs and MAb analogs and different surface-area-to-volume ratios. Doing so should provide us with a further understanding of differences among metal containers both immediately after exposure to metal contact surfaces under ambient conditions and after subsequent exposure to thermal stress in final drug-product containers.
Acknowledgments
The authors thank members of the Regeneron quality control analytical sciences team, including Stacey Helming, and Andrea Smith; members of therapeutic proteins team, including Cristinel Sandu, Humam-Al-Rubaye and Mushhood Sheikh; members of the analytical chemistry group, including Shivkumar Raidas and Xuefei Zhong; and members of the drug product process development team, including Gorgi Pavlov, Gitanjali Sharma, Trinh Vo, and Bailey Cooper for assisting with analytical testing.
References
1 Bee JS, et al. Aggregation of a Monoclonal Antibody Induced By Adsorption to Stainless Steel. Biotechnol. Bioeng. 105(1) 2010: 121–129; https://doi.org/10.1002/bit.22525.
2 Kalonia CK, et al. Protein Adsorption and Layer Formation at the Stainless Steel-Solution Interface Mediates Shear-Induced Particle Formation for an IgG1 Monoclonal Antibody. Mol. Pharm. 15(3) 2018: 1319–1331; https://doi.org/10.1021/acs.molpharmaceut.7b01127.
3 Zhou S, Schöneich C, Singh S. Biologics Formulation Factors Affecting Metal Leachables from Stainless Steel. AAPS PharmSciTech. 12(1) 2011: 411–421; https://doi.org/10.1208/s12249-011-9592-3.
4 Zhou S, et al. Biotherapeutic Formulation Factors Affecting Metal Leachables from Stainless Steel Studied by Design of Experiments. AAPS PharmSciTech. 13(1) 2012: 284–294; https://doi.org/10.1208/s12249-011-9747-2.
5 Zhou S, Lewis LM, Singh S. Metal Leachables in Therapeutic Biologic Products: Origin, Impact, and Detection. Am. Pharm. Rev. 13, 2010: 76–80.
6 Lam XM, Yang JY, Cleland JL. Antioxidants for Prevention of Methionine Oxidation in Recombinant Monoclonal Antibody HER2. J. Pharm. Sci. 86(11) 1997: 1250–1255; https://doi.org/10.1021/js970143s.
7 Chen B, et al. Influence of Histidine on the Stability and Physical Properties of a Fully Human Antibody in Aqueous and Solid Forms. Pharm. Res. 20(12) 2003: 1952–1960; https://doi.org/10.1023/b:pham.0000008042.15988.c0.
8 Grillo AO. Late-State Formulation Development and Characterization of Biopharmaceuticals. Jameel F, Hershenson S, Eds. Formulation and Process Development Strategies for Manufacturing Biopharmaceuticals. John Wiley & Sons: Hoboken, NJ, 2010: 161–171.
9 Klair N, et al. Stress Temperature Studies in Small Scale Hastelloy Drug Substance Containers Lead to Increased Extent of and Increased Variability in Antibody–Drug Conjugate and Monoclonal Antibody Aggregation: Evidence for Novel Oxidation-Induced Crosslinking in Monoclonal Antibodies. J. Pharm Sci. 110(4) 2021: 1615–1624; https://doi.org/10.1016/j.xphs.2020.09.052.
Corresponding author Sonia Bedi ([email protected]) is associate director and Mintai Peter Hwang is a scientist in the formulation development group; Yunlong Zhao is a staff scientist, and Yuan Mao is associate director in the analytical chemistry group; Amardeep Singh Bhalla is senior director in the formulation development group; Ning Li is executive director in the analytical chemistry group; and Mohammed Shameem is vice president in the formulation development group, all at Regeneron Pharmaceuticals Inc., 777 Old Saw Mill River Road, Tarrytown, NY 10591.
The authors declare no conflict of interest.
You May Also Like






