May 2022 Featured Report
https://stock.adobe.com
Critical to the manufacturing of biotherapeutics is development of robust and stable cell lines that express high-quality products in large quantities. Chinese hamster ovary (CHO) cells are the “workhorse” expression host for manufacturing glycoprotein-based therapeutics — from antibodies to enzymes and hormones and beyond — making them the subject of most industry discussion regarding both cell-line development (CLD) and engineering. Most methods, technologies, and strategies apply regardless of the type of protein to be expressed or the type of cells expressing it.
On 15–17 March 2022, Informa Connect Life Sciences presented the main BioProcess International US West Conference and Exhibition program live in San Diego, CA. That was followed the next week with online-only roundtable discussions on key topics from the in-person sessions. Here I report on the main themes and some key presentations from the track that addressed the latest developments in CLD. What stood out most to me...
https://www.alamy.com
At BPI Europe in April 2019, Dirk Gewert (business unit leader of bioproduction at Horizon Discovery) told
BioProcess Insider
that Chinese hamster ovary (CHO) cell lines haven’t changed much in the 30 years since they were first used in biomanufacturing (
1
). Only a few companies offered commercially available, production-ready CHO cell lines for large-scale manufacturing of biotherapeutics. “In all cases,” he said, “cell lines were selected by identifying high-expressing clones and focusing on process optimization to improve expression and other critical quality attributes.”
Gewart’s company launched a consortium of suppliers to improve that situation. Its key objective was “to leverage modern gene-editing tools to improve the biology at the heart of the cell, ultimately to generate a fundamentally improved CHO expression platform available to the entire industry for the manufacturing of routine biotherapeutics.” Using genome-editing tools such as clustered regularly interspaced ...
The polyvalency of IgM molecules gives them high-avidity binding to their targets.
HTTPS://IGMBIO.COM
Figure 1:
Compare the structures of IgG and IgM antibodies.
Immunoglobulin G (IgG) antibodies have been studied and applied as biopharmaceuticals for decades, and they remain dominant in the monoclonal antibody (MAb) pipeline. By contrast, immunoglobulin M (IgM) molecules are much larger and consequently more challenging for biomanufacturing and therapeutic application. Essentially, they appear as clusters of the familiar Y-shaped IgG molecules, joined at their bases in pentameric (Figure 1) or hexameric forms. That structure gives them 10 and 12 binding moieties, respectively, which translate to superior binding power (
avidity
) of the antibodies compared with IgG isomers.
IGM Biosciences of Mountain View, CA, believes that IgMs could be more effective than IgGs at treating certain diseases — in particular as cancer immunotherapies. Many researchers in the past have been stymied by the technical barri...
A number of factors contribute to delivering a robust, highly productive, and reliable process for manufacturing a therapeutic protein. They begin with a cell-host system and a gene-expression strategy that determine a developer’s ability to optimize growth and expression titers. But for many therapeutic proteins, initial attempts to develop a production process are based on evaluation of limited factors and tend to yield only small quantities or poor product quality. Automation can enable parallel building and expression screening of diverse expression strategies within a robust host cell system, accelerating product development. Ensuring significant production of a therapeutic protein from the start, and thus quickly generating high protein quality and yield, can provide significant advantages in opportunity costs.
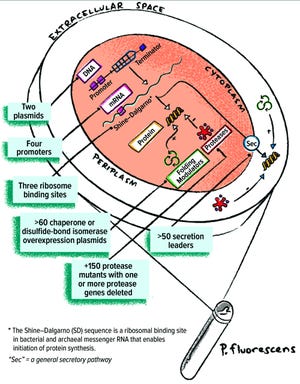
Formerly known as
Pfenex
expression technology, the Pelican Expression Technology platform (PET) is a microbial expression system based on the Gram-negative bacterium
Pseudomonas fluoresce...
Cell-line development (CLD) involves screening thousands of clones to identify the most stable and productive candidate for an upstream manufacturing process (Figure 1). Such assessment requires considerable time and resources for preparation and execution of multiple cultivations and analytical assays. In March 2022, Lukas Klein and Dirk Müller (scientist and manager of media and process development, respectively, at Sartorius) delivered a BPI Ask the Expert webinar about integrating their company’s Ambr 15 Cell Culture bioreactor and Octet label-free biolayer interferometry (BLI) technology to accelerate CLD studies. The presenters highlighted applications for media screening and platform optimization.
Figure 1:
Cell-line development (CLD) comprises several activities that require extensive time, resources, and analytical capabilities. The BPI Ask the Expert presentation summarized in this article focused on technologies for streamlining studies in steps two, three, and four of the CLD process, as outl...
Subscribe to receive our monthly print or digital publication
Join our 70,000+ readers. And yes, it's completely free.