October 2020 Featured Report
https://stock.adobe.com
Still the largest sector of the industry, monoclonal antibodies (MAbs) have dominated the biopharmaceutical stage for over 30 years. Some observers might think there’s nothing new to say about these molecules; others point to antibody derivatives as a more exciting alternative. But MAbs are far from an outdated technology. From biosimilar developments to cell-free synthesis to yeast display, immunogenicity improvements, and fully human antibodies — as well as improvements in process efficiency and cost reductions, as discussed herein — the MAb future not only is bright, but also could be amazing.
Our fellow Informa publication, the journal
mAbs
, started out this year with a cost-benchmarking study from authors at University College London and AstraZeneca (
1
). Farid et al. analyzed the contribution of process development and manufacturing to overall research and development (R&D) costs. The authors’ model captured cost, time, and risk and took into account the interdependencies ...
Antibodies attack invaders such as the SARS-CoV-2 virus that causes COVID-19 illness.
Commercial development of therapeutic monoclonal antibodies (MAbs) began in the early 1980s, and by 1986 the first MAb product had been approved in the United States: muromonab-CD3 (trade name Orthoclone OKT3, marketed by Janssen-Cilag) for prevention of kidney-transplant rejection. Since its approval, therapeutic MAbs and antibody-related products such as Fc-fusion proteins, antibody fragments, and antibody–drug conjugates (collectively referred to herein as “MAb products”) have grown to become the dominant product class within the biopharmaceutical market. They have been approved for treating a broad range of of diseases, from those with patient populations of a few thousand or less (e.g., orphan indications such as hemophagocytic lymphohistiocytosis) to those afflicting hundreds of thousands (e.g., some cancers and multiple sclerosis) or even millions of patients (diseases such as asthma and rheumatoid arthritis). Wit...
The COVID-19 pandemic has generated a significant and rapid response from scientists who aim to develop drugs and vaccines in the academic, government, and industrial sectors. Such interventions are essential to containing SARS-CoV-2, the coronavirus that causes the COVID-19 disease. To inform and educate the public about global efforts to develop targeted therapies such as monoclonal antibodies (MAbs), The Antibody Society (TAS) and the Chinese Antibody Society (CAS) have designed and implemented a freely available online database called the COVID-19 Antibody Therapeutics Tracker (
1
).
Janice Reichert (executive director of TAS and founding editor in chief of the Informa Taylor & Francis journal
mAbs
) recalls the moment when she realized the enormity of charting the therapeutic antibody industry’s response to the COVID-19 pandemic: “I’ve tracked trends in the development of antibody therapeutics for over 20 years. The first paper that I wrote on the topic was published in 2001. So tracking COVID-19 in...
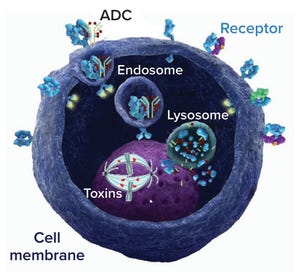
Antibody–drug conjugate (ADC) mechanism of action; an ADC binds to a cell-surface target receptor, which is internalized to form an endosome and merge with a cell lysosome. Drug is released from the lysosome and acts on its target (DNA or microtubule) to kill the cell.
G-protein–coupled receptors (GPCRs) are a large and diverse family of seven-transmembrane–domain proteins expressed on the surface of human cells. These molecules respond to external stimuli by initiating signal-transduction pathways that affect the expression of a large family of genes — which, in turn, regulate a range of vital physiological processes and functions. Figure 1 illustrates the general pathways of GPCRs.
Without these proteins, humans simply could not survive: Without β-adrenergic receptors, we could not regulate our blood sugar, for example; and without serotonin receptors, we could not experience happiness. Many physiological functions are related to the signaling pathways regulated by GPCRs, and dysfunctions of those pathw...
Subscribe to receive our monthly print or digital publication
Join our 70,000+ readers. And yes, it's completely free.