September 2021 Featured Report
Restricted-access technology is used at Grand River Aseptic Manufacturing’s large-scale fill–finish site in Grand Rapids, MI. It was awarded the 2021 Facility of the Year award for operational agility related to COVID-19 by the International Society for Pharmaceutical Engineering (ISPE). grand river aseptic manufacturing (
https://www.grandriverasepticmfg.com
)
Biological drug substances are constituent in a wide range of medicinal products with an even broader spectrum of applications. Those include autoimmune-disease treatments (e.g., for arthritis), vaccines, and recombinant therapeutic proteins (e.g., for cancer treatment). What such products all have in common is that they are manufactured using biotechnology and other cutting-edge technologies. Biologics are not as physically robust as their small-molecule counterparts. Hence, during biomanufacturing processes, these complex molecules present a number of challenges. Some of the typical shared characteristics of biopharmaceuticals include
During down...
The development of new medicines is a highly regulated process focused on demonstrating efficacy and safety of new products. Although such qualities always will remain the primary focus of drug development, the biopharmaceutical industry gradually has adopted additional design aspects. New approaches can help meet patients’ divergent needs to improve their lives in meaningful ways.
Often referred to as “patient-centered” or “patient-focused” drug-product design, such considerations are expected by many experts to become an increasingly dominant part of future drug development. Patient centricity presents in many different ways, all generally relating to convenience and safety of administration, patient compliance with treatment, and general patient engagement. No single or universal solution applies to all disease conditions and all patients. Each patient group, condition, and treatment paradigm requires a different patient-centered approach to maximize compliance, safety, and treatment outcomes.
A number...
milliporesigma (https://www.emdmillipore.com)
Polysorbate-80 (PS-80) and polysorbate-20 (PS-20) are used widely in formulation of biotherapeutic products for preventing surface adsorption and as stabilizers against protein aggregation (
1
). Degradation of polysorbates can cause turbidity and potential formation of subvisible particles mainly consisting of poorly soluble hydrophobic free fatty acids (
1
). Polysorbate degradation is an industry-wide challenge both in biotherapeutics processing and formulation development. The risk of such degradation increases with higher cell densities and greater expression titers in bioprocessing, as well as with higher drug concentration in biologic formulations (
2
–
3
).
The main mechanisms for polysorbate degradation are oxidation and hydrolysis (
1
). Observations of temperature-dependent degradation that does not occur in placebo controls have led many experts to conclude that enzymatic hydrolysis is the major root cause of polysorbate degradation in biotherapeut...
How can biopharmaceutical manufacturers expect their suppliers to deliver what the industry wants and needs if it doesn’t communicate those desires? Without industry input, biotechnology suppliers are developing technologies “blindly.” Despite their having delivered great value over the years, together the greater community can do better. Aligned industry input is a vital element in the development process. Here we describe how two BioPhorum workstreams focused on drug-product development have worked to facilitate these communications.
Last summer, the container–closure integrity (CCI) workstream gave advance notice in a BPI eBook of its planned suite of papers describing how a quality- and risk-based holistic approach could remove the need for routine CCI testing (
1
). Keeping unnecessary testing burdens to a minimum enhances manufacturing efficiencies and maintains patient supplies through building quality into products and processes — rather than fixating on a “testing into compliance” mindset.
The ho...
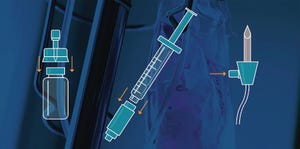
Closed-system transfer devices facilitate preparation and administration of precise
injectable doses. (reference
3
)
Ever since the first biopharmaceutical product (biologic) was approved in the 1980s, companies have developed protocols and tests to ensure that such products are safe and effective. Biologics are very different from traditional small-molecule drugs, with unique risks inherent to their manufacturing processes. Biopharmaceutical formulations often present as complex mixtures that can be sensitive to heat, light, and many other factors, all of which must be monitored and assessed. However, until recently, developers worked mostly independently, with only their own resources and expertise to develop those assessments and set acceptable parameters.
BioPhorum recognized that that competing companies could work together and share information in many ways that would accelerate progress for everyone. We identified two areas in which this was especially timely: compatibility assessments of biopharm...
Subscribe to receive our monthly print or digital publication
Join our 70,000+ readers. And yes, it's completely free.