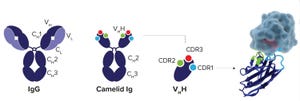
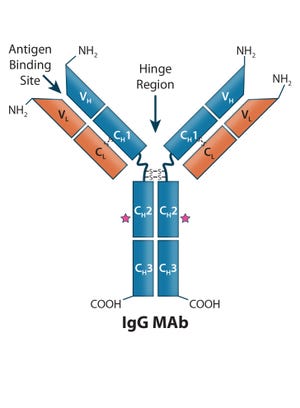
Purification of Hepatitis B Virus Surface Antigen for Vaccine Products: Impact of Ligand Density on HBsAg Purification By Immunoaffinity
Vaccines
Purification of Hepatitis B Virus Surface Antigen for Vaccine Products: Impact of Ligand Density on HBsAg Purification By Immunoaffinity ChromatographyPurification of Hepatitis B Virus Surface Antigen for Vaccine Products: Impact of Ligand Density on HBsAg Purification By Immunoaffinity Chromatography