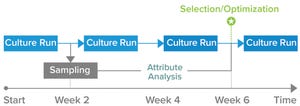
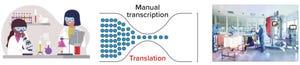
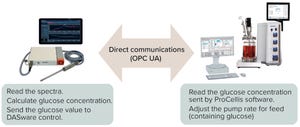
Process Monitoring and Controls
Cleaning Validation Acceptance Limits for Biological Process Residues: Part 1 — Acceptable Exposure of Degraded Proteins Based on Reference
Validation
Cleaning Validation Acceptance Limits for Biological Process Residues: Part 1 — Acceptable Exposure of Degraded Proteins Based on Reference ImmunogensCleaning Validation Acceptance Limits for Biological Process Residues: Part 1 — Acceptable Exposure of Degraded Proteins Based on Reference Immunogens